Formulation and in vitro-in vivo evaluation of black raspberry extract-loaded PLGA/PLA injectable millicylindrical implants for sustained delivery of chemopreventive anthocyanins
- PMID: 20148292
- PMCID: PMC2880396
- DOI: 10.1007/s11095-009-0038-5
Formulation and in vitro-in vivo evaluation of black raspberry extract-loaded PLGA/PLA injectable millicylindrical implants for sustained delivery of chemopreventive anthocyanins
Abstract
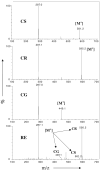
Purpose: The objective of this study was to formulate and evaluate freeze-dried black raspberry (FBR) ethanol extract (RE) loaded poly(DL-lactic-co-glycolic acid) (PLGA) and poly(DL-lactic acid) (PLA) injectable millicylindrical implants for sustained delivery of chemopreventive FBR anthocyanins (cyanidin-3-sambubioside (CS), cyanidin-3-glucoside (CG) and cyanidin-3-rutinoside (CR)).
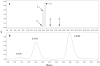
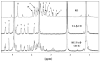
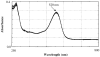
Methods: Identification and quantitation of CS, CG, and CR in RE was performed by mass spectroscopy and HPLC. RE:triacetyl-beta-cyclodextrin (TA-beta-CD) inclusion complex (IC) was prepared by a kneading method and characterized by X-ray diffraction (XRD), nuclear magnetic resonance spectroscopy (NMR) and UV-visible spectroscopy. RE or RE:TA-beta-CD IC-loaded PLGA or PLA implants were prepared by a solvent extrusion method. In vitro and in vivo controlled release studies were conducted in phosphate-buffered saline Tween-80 (pH 7.4, 37 degrees C) and after subcutaneous administration in male Sprague-Dawley rats, respectively. Anthocyanins were quantified by HPLC at 520 nm.
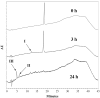
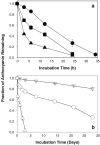
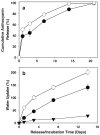
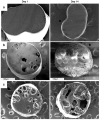
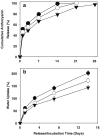
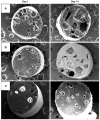
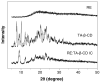
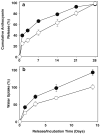
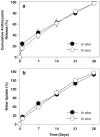
Results: The content of CS, CG, and CR in RE was 0.2, 1.5, and 3.5 wt%, respectively. The chemical stability of anthocyanins in solution was determined to be pH-dependent, and their degradation rate increased with an increase in pH from 2.4 to 7.4. PLGA/PLA millicylindrical implants loaded with 5 or 10 wt% RE exhibited a high initial burst and short release duration of anthocyanins (35-52 and 80-100% CG + CR release after 1 and 14 days, respectively). The cause for rapid anthocyanins release was linked to higher polymer water uptake and porosity associated with the high osmolytic components of large non-anthocyanin fraction of RE. XRD, (1)H NMR and UV-visible spectroscopy indicated that the non-anthocyanin fraction molecules of RE formed an IC with TA-beta-CD, decreasing the hydrophilicity of RE. Formation of an IC with hydrophobic carrier, TA-beta-CD, provided better in vitro/in vivo sustained release of FBR anthocyanins (16-24 and 97-99% CG + CR release, respectively, after 1 and 28 days from 20 wt% RE:TA-beta-CD IC/PLA implants) over 1 month, owing to reduced polymer water uptake and porosity.
Conclusion: PLA injectable millicylindrical implants loaded with RE:TA-beta-CD IC are optimal dosage forms for 1-month slow and continuous delivery of chemopreventive FBR anthocyanins.
Figures

Similar articles
-
Formulation and characterization of injectable poly(DL-lactide-co-glycolide) implants loaded with N-acetylcysteine, a MMP inhibitor.Pharm Res. 2008 Mar;25(3):586-97. doi: 10.1007/s11095-007-9430-1. Epub 2007 Sep 22. Pharm Res. 2008. PMID: 17891553 Free PMC article.
-
Effect of formulation parameters on 2-methoxyestradiol release from injectable cylindrical poly(DL-lactide-co-glycolide) implants.Eur J Pharm Biopharm. 2008 Sep;70(1):187-98. doi: 10.1016/j.ejpb.2008.03.007. Epub 2008 Mar 20. Eur J Pharm Biopharm. 2008. PMID: 18472254 Free PMC article.
-
Influence of the microencapsulation method and peptide loading on poly(lactic acid) and poly(lactic-co-glycolic acid) degradation during in vitro testing.J Control Release. 1998 Feb 12;51(2-3):327-41. doi: 10.1016/s0168-3659(97)00188-0. J Control Release. 1998. PMID: 9685930
-
Measuring erosion of biodegradable polymers in brimonidine drug delivery implants by quantitative proton NMR spectroscopy (q-HNMR).J Pharm Sci. 2025 Jan;114(1):245-255. doi: 10.1016/j.xphs.2024.08.028. Epub 2024 Aug 31. J Pharm Sci. 2025. PMID: 39218154
-
Recent advances in the preparation progress of protein/peptide drug loaded PLA/PLGA microspheres.Yao Xue Xue Bao. 2007 Jan;42(1):1-7. Yao Xue Xue Bao. 2007. PMID: 17520799 Review.
Cited by
-
Investigating PLGA microparticle swelling behavior reveals an interplay of expansive intermolecular forces.Sci Rep. 2021 Jul 15;11(1):14512. doi: 10.1038/s41598-021-93785-6. Sci Rep. 2021. PMID: 34267274 Free PMC article.
-
Controlled-release systemic delivery - a new concept in cancer chemoprevention.Carcinogenesis. 2012 Aug;33(8):1608-15. doi: 10.1093/carcin/bgs209. Epub 2012 Jun 13. Carcinogenesis. 2012. PMID: 22696595 Free PMC article.
-
Optimizing therapeutic efficacy of chemopreventive agents: A critical review of delivery strategies in oral cancer chemoprevention clinical trials.J Carcinog. 2011;10:23. doi: 10.4103/1477-3163.85185. Epub 2011 Sep 21. J Carcinog. 2011. PMID: 22013393 Free PMC article.
-
The Effect of Polymer Blends on the In Vitro Release/Degradation and Pharmacokinetics of Moxidectin-Loaded PLGA Microspheres.Int J Mol Sci. 2023 Sep 29;24(19):14729. doi: 10.3390/ijms241914729. Int J Mol Sci. 2023. PMID: 37834176 Free PMC article.
-
PLGA Implants for Controlled Drug Delivery and Regenerative Medicine: Advances, Challenges, and Clinical Potential.Pharmaceuticals (Basel). 2025 Apr 27;18(5):631. doi: 10.3390/ph18050631. Pharmaceuticals (Basel). 2025. PMID: 40430452 Free PMC article. Review.
References
-
- Jemal A, Siegel R, Ward E, Hao YP, Xu JQ, Murray T, et al. Cancer statistics. CA-Cancer J Clin. 2008;58:71–96. - PubMed
-
- Braakhuis BJM, Tabor MP, Leemans CR, van der Waal I, Snow GB, Brakenhoff RH. Second primary tumors and field cancerization in oral and oropharyngeal cancer: molecular techniques provide new insights and definitions. Head Neck-J Sci Spec. 2002;24:198–206. - PubMed
-
- Malone JP, Stephens JA, Grecula JC, Rhoades CA, Ba BAG, Ghaheri BA, et al. Disease control, survival, and functional outcome after multimodal treatment for advanced-stage tongue base cancer. Head Neck-J Sci Spec. 2004;26:561–72. - PubMed
-
- Pathak KA, Gupta S, Talole S, Khanna V, Chaturvedi P, Deshpande MS, et al. Advanced squamous cell carcinoma of lower gingivobuccal complex: patterns of spread and failure. Head Neck-J Sci Spec. 2005;27:597–602. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

