Lovastatin inhibits the thrombin-induced loss of barrier integrity in bovine corneal endothelium
- PMID: 20148651
- PMCID: PMC3096541
- DOI: 10.1089/jop.2009.0025
Lovastatin inhibits the thrombin-induced loss of barrier integrity in bovine corneal endothelium
Abstract
Purpose: Increased actomyosin contraction of the dense band of actin cytoskeleton at the apical junctional complex (perijunctional actomyosin ring, PAMR) breaks down the barrier integrity of corneal endothelium. This study has investigated the efficacy of statins, which inhibit activation of RhoA, in opposing the thrombin-induced loss of barrier integrity of monolayers of cultured bovine corneal endothelium.
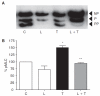
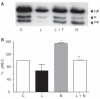
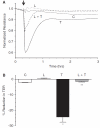
Methods: Myosin light chain (MLC) phosphorylation, a biochemical measure of actomyosin contraction, was assayed by urea-glycerol gel electrophoresis, followed by western blot analysis. The locus of MLC phosphorylation and changes in the organization of the PAMR were visualized by immunostaining. Phosphorylation of MYPT1, a regulatory subunit of myosin light-chain phosphatase (MLCP), was assessed by Western blot analysis to determine down-regulation of RhoA. The barrier integrity was assessed in terms of trans-endothelial electrical resistance (TER), and further confirmed by determining permeability to FITC dextran (10 kDa) and distribution of ZO-1, a marker of tight junctional assembly.
Results: Lovastatin, a prototype of lipophilic statins, induced MLC dephosphorylation under basal conditions. It opposed increase in phosphorylation of MLC and MYPT1 in response to thrombin and nocodazole, agents known to activate RhoA in the endothelium. Pretreatment with the statin opposed the thrombin- and nocodazole-induced disruption of the PAMR and the thrombin-induced decline in TER. Lovastatin also opposed the thrombin- and nocodazole-induced increase in permeability to FITC dextran and redistribution of ZO-1. However, upon supplementation with GGPP (geranylgeranyl pyrophosphate), lovastatin failed to oppose the effects of thrombin and nocodazole on the PAMR, ppMLC, and ZO-1 distribution.
Conclusions: Lovastatin attenuates RhoA activation in the corneal endothelium presumably by reducing its isoprenylation. This underlies the suppression of the thrombin-induced loss in barrier integrity of the corneal endothelium.
Figures








Similar articles
-
Microtubule disassembly breaks down the barrier integrity of corneal endothelium.Exp Eye Res. 2009 Sep;89(3):333-43. doi: 10.1016/j.exer.2009.03.019. Epub 2009 Apr 2. Exp Eye Res. 2009. PMID: 19345211 Free PMC article.
-
Thrombin-induced phosphorylation of the regulatory light chain of myosin II in cultured bovine corneal endothelial cells.Exp Eye Res. 2004 Oct;79(4):477-86. doi: 10.1016/j.exer.2004.06.018. Exp Eye Res. 2004. PMID: 15381032
-
Formation and disassembly of adherens and tight junctions in the corneal endothelium: regulation by actomyosin contraction.Invest Ophthalmol Vis Sci. 2010 Apr;51(4):2139-48. doi: 10.1167/iovs.09-4421. Epub 2009 Dec 17. Invest Ophthalmol Vis Sci. 2010. PMID: 20019371 Free PMC article.
-
Cell signaling in regulation of the barrier integrity of the corneal endothelium.Exp Eye Res. 2012 Feb;95(1):8-15. doi: 10.1016/j.exer.2011.09.009. Epub 2011 Sep 24. Exp Eye Res. 2012. PMID: 21963716 Free PMC article. Review.
-
Dynamic regulation of barrier integrity of the corneal endothelium.Optom Vis Sci. 2010 Apr;87(4):E239-54. doi: 10.1097/OPX.0b013e3181d39464. Optom Vis Sci. 2010. PMID: 20142793 Free PMC article. Review.
Cited by
-
Elevated cytokines, thrombin and PAI-1 in severe HCPS patients due to Sin Nombre virus.Viruses. 2015 Feb 10;7(2):559-89. doi: 10.3390/v7020559. Viruses. 2015. PMID: 25674766 Free PMC article.
-
Establishment of the deuterium oxide dilution method as a new possibility for determining the transendothelial water permeability.Pflugers Arch. 2024 Jun;476(6):993-1005. doi: 10.1007/s00424-024-02934-z. Epub 2024 Mar 5. Pflugers Arch. 2024. PMID: 38438679 Free PMC article.
-
Evaluation of the effect of atorvastatin on corneal endothelial cells during the initial 12-month period after acute coronary syndrome.Indian J Ophthalmol. 2024 Aug 1;72(8):1142-1146. doi: 10.4103/IJO.IJO_3201_23. Epub 2024 Jul 29. Indian J Ophthalmol. 2024. PMID: 39078958 Free PMC article.
-
Inhibition of Rho and Rac geranylgeranylation by atorvastatin is critical for preservation of endothelial junction integrity.PLoS One. 2013;8(3):e59233. doi: 10.1371/journal.pone.0059233. Epub 2013 Mar 13. PLoS One. 2013. PMID: 23555637 Free PMC article.
-
Barrier stabilizing mediators in regulation of microvascular endothelial permeability.Chin J Traumatol. 2012;15(2):105-12. doi: 10.3760/cma.j.issn.1008-1275.2012.02.008. Chin J Traumatol. 2012. PMID: 22480675 Free PMC article. Review.
References
-
- Noske W., Fromm M., Levarlet B., et al. Tight junctions of the human corneal endothelium: morphological and electrophysiological features. Ger. J. Ophthalmol. 1994;3:253–257. - PubMed
-
- Srinivas S.P., Satpathy M., Gallagher P., et al. Adenosine induces dephosphorylation of myosin II regulatory light chain in cultured bovine corneal endothelial cells. Exp. Eye Res. 2004;79:543–551. - PubMed
-
- Edelhauser H.F. The balance between corneal transparency and edema: the Proctor Lecture. Invest. Ophthalmol. Vis. Sci. 2006;47:1754–1767. - PubMed
-
- Fischbarg J., Lim J.J. Fluid and electrolyte transports across corneal endothelium. Curr. Top. Eye Res. 1984;4:201–223. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

