Coordinate action of membrane-type matrix metalloproteinase-1 (MT1-MMP) and MMP-2 enhances pericellular proteolysis and invasion
- PMID: 20148894
- PMCID: PMC11158779
- DOI: 10.1111/j.1349-7006.2010.01498.x
Coordinate action of membrane-type matrix metalloproteinase-1 (MT1-MMP) and MMP-2 enhances pericellular proteolysis and invasion
Abstract
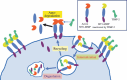
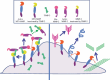
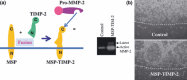
Membrane-type matrix metalloproteinase-1 (MT1-MMP) mediates cleavage of not only MMP-2/gelatinase A for activation, but also a variety of substrates including type I collagen (reviewed in Cancer Sci 2005; 96: 212-7). MMP-2 activation involves tissue inhibitor of MMP (TIMP)-2 as a bridging molecule between MT1-MMP and pro-MMP-2. Thus, net activity of MT1-MMP and MMP-2 is regulated in a complex manner depending on TIMP-2 concentration. During invasive growth of tumor cells in type I collagen matrix, MT1-MMP initiates denaturation of collagen into gelatin, which is subsequently digested further by MMP-2 adjacent to MT1-MMP. Coordinate action of MT1-MMP and MMP-2 may facilitate pericellular proteolysis, and enhance not only tumor invasion/migration but also cell growth. Tetraspanins as binding proteins of MT1-MMP regulate MT1-MMP subcellular localization and compartmentalization, leading to efficient MMP-2 activation and proteolysis coupled with cellular function.
Figures

Similar articles
-
TIMP-2 (tissue inhibitor of metalloproteinase-2) regulates MMP-2 (matrix metalloproteinase-2) activity in the extracellular environment after pro-MMP-2 activation by MT1 (membrane type 1)-MMP.Biochem J. 2003 Sep 15;374(Pt 3):739-45. doi: 10.1042/BJ20030557. Biochem J. 2003. PMID: 12755684 Free PMC article.
-
Roles of membrane type 1 matrix metalloproteinase and tissue inhibitor of metalloproteinases 2 in invasion and dissemination of human malignant glioma.J Neurosurg. 2001 Mar;94(3):464-73. doi: 10.3171/jns.2001.94.3.0464. J Neurosurg. 2001. PMID: 11235952
-
Tissue inhibitor of matrix metalloproteinase-2 regulates matrix metalloproteinase-2 activation by modulation of membrane-type 1 matrix metalloproteinase activity in high and low invasive melanoma cell lines.J Biol Chem. 1999 Jul 23;274(30):21056-62. doi: 10.1074/jbc.274.30.21056. J Biol Chem. 1999. PMID: 10409657
-
MT1-MMP: a potent modifier of pericellular microenvironment.J Cell Physiol. 2006 Jan;206(1):1-8. doi: 10.1002/jcp.20431. J Cell Physiol. 2006. PMID: 15920734 Review.
-
Roles of pericellular proteolysis by membrane type-1 matrix metalloproteinase in cancer invasion and angiogenesis.Cancer Sci. 2003 Jul;94(7):569-74. doi: 10.1111/j.1349-7006.2003.tb01484.x. Cancer Sci. 2003. PMID: 12841863 Free PMC article. Review.
Cited by
-
Regulation of ligands for the NKG2D activating receptor.Annu Rev Immunol. 2013;31:413-41. doi: 10.1146/annurev-immunol-032712-095951. Epub 2013 Jan 3. Annu Rev Immunol. 2013. PMID: 23298206 Free PMC article. Review.
-
The extracellular matrix in tumor progression and metastasis.Clin Exp Metastasis. 2019 Jun;36(3):171-198. doi: 10.1007/s10585-019-09966-1. Epub 2019 Apr 11. Clin Exp Metastasis. 2019. PMID: 30972526 Review.
-
Functional interplay between tetraspanins and proteases.Cell Mol Life Sci. 2011 Oct;68(20):3323-35. doi: 10.1007/s00018-011-0746-y. Epub 2011 Jun 18. Cell Mol Life Sci. 2011. PMID: 21687991 Free PMC article. Review.
-
Matrix metalloproteinase-9-null mice are resistant to TGF-β-induced anterior subcapsular cataract formation.Am J Pathol. 2014 Jul;184(7):2001-12. doi: 10.1016/j.ajpath.2014.03.013. Epub 2014 May 6. Am J Pathol. 2014. PMID: 24814605 Free PMC article.
-
New Derivatives of N-Hydroxybutanamide: Preparation, MMP Inhibition, Cytotoxicity, and Antitumor Activity.Int J Mol Sci. 2023 Nov 15;24(22):16360. doi: 10.3390/ijms242216360. Int J Mol Sci. 2023. PMID: 38003553 Free PMC article.
References
-
- Birkedal HH, Moore WG, Bodden MK et al. Matrix metalloproteinases: a review . Crit Rev Oral Biol Med 1993; 4: 197–250. - PubMed
-
- Stetler‐Stevenson WG, Aznavoorian S, Liotta LA. Tumor cell interactions with the extracellular matrix during invasion and metastasis. Annu Rev Cell Biol 1993; 9: 541–73. - PubMed
-
- Matrisian LM. Metalloproteinases and their inhibitors in matrix remodeling. Trends Genet 1990; 6: 121–5. - PubMed
-
- Curry TJ, Mann JS, Estes RS, Jones PB. Alpha 2‐macroglobulin and tissue inhibitor of metalloproteinases: collagenase inhibitors in human preovulatory ovaries. Endocrinology, 1990; 127: 63–8. - PubMed
-
- Zhu C, Woessner JJ. A tissue inhibitor of metalloproteinases and α‐macroglobulins in the ovulating rat ovary: possible regulators of collagen matrix breakdown. Biol Reprod 1991; 45: 334–42. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

