A high-affinity putrescine-cadaverine transporter from Trypanosoma cruzi
- PMID: 20149109
- PMCID: PMC3183094
- DOI: 10.1111/j.1365-2958.2010.07081.x
A high-affinity putrescine-cadaverine transporter from Trypanosoma cruzi
Abstract
Whereas mammalian cells and most other organisms can synthesize polyamines from basic amino acids, the protozoan parasite Trypanosoma cruzi is incapable of polyamine biosynthesis de novo and therefore obligatorily relies upon putrescine acquisition from the host to meet its nutritional requirements. The cell surface proteins that mediate polyamine transport into T. cruzi, as well as most eukaryotes, however, have by-in-large eluded discovery at the molecular level. Here we report the identification and functional characterization of two polyamine transporters, TcPOT1.1 and TcPOT1.2, encoded by alleles from two T. cruzi haplotypes. Overexpression of the TcPOT1.1 and TcPOT1.2 genes in T. cruzi epimastigotes revealed that TcPOT1.1 and TcPOT1.2 were high-affinity transporters that recognized both putrescine and cadaverine but not spermidine or spermine. Furthermore, the activities and subcellular locations of both TcPOT1.1 and TcPOT1.2 in intact parasites were profoundly influenced by extracellular putrescine availability. These results establish TcPOT1.1 and TcPOT1.2 as key components of the T. cruzi polyamine transport pathway, an indispensable nutritional function for the parasite that may be amenable to therapeutic manipulation.
Figures
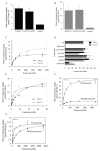
 ) that had been grown in SDM-79 containing 10% FBS and 200 μM putrescine. Values for endogenous transport in control pTEX-TcGRASP::GFP- or pTEX-GFP-transfected parasites were subtracted from the experimental rates. D. Inhibition profile for TcPOT1.1- and TcPOT1.2-mediated putrescine transport. The ability of the pTEX-TcPOT1.1::GFP and pTEX-TcPOT1.2::GFP transfectants to take up 0.5 μM [3H]putrescine was evaluated in the presence of a variety of structurally related non-radiolabelled compounds, each at a concentration of 50 μM. Results are plotted as a percentage of putrescine uptake obtained without inhibitor (NI). Each value represents the mean ± SD of three independent experiments. E. Abilities of epimastigotes transfected with either pTEX-TcPOT1.1::GFP or pTEX-TcPOT1.2::GFP and grown in putrescine-supplemented SDM-79 medium to take up [14C]cadaverine over a range of cadaverine concentrations. Uptake rates obtained with the control pTEX-TcGRASP::GFP transfectant were subtracted from experimental data points. Data were fitted to a Michaelis-Menten algorithm to determine apparent Km and Vmax values. F. Michaelis-Menten plot of putrescine uptake rates obtained for T. cruzi epimastigotes expressing pTEX-TcPOT1.1::GFP that had been grown either in SDM-79 containing 10% FBS in the absence (○) or presence (●) of 200 μM putrescine for 48 h. The pTEX-TcGRASP::GFP expressing transfectant served as the control for endogenous transport, and control rates were subtracted from the experimental rates. G. Michaelis-Menten analysis of putrescine uptake rates obtained for T. cruzi epimastigotes expressing pTEX-TcPOT1.2::GFP that had been grown either in SDM-79 containing 10% FBS in the absence (○) or presence (●) of 200 μM putrescine for 48 h. The pTEX-GFP expressing transfectant served as the control for endogenous transport and control rates were subtracted from the experimental rates.
) that had been grown in SDM-79 containing 10% FBS and 200 μM putrescine. Values for endogenous transport in control pTEX-TcGRASP::GFP- or pTEX-GFP-transfected parasites were subtracted from the experimental rates. D. Inhibition profile for TcPOT1.1- and TcPOT1.2-mediated putrescine transport. The ability of the pTEX-TcPOT1.1::GFP and pTEX-TcPOT1.2::GFP transfectants to take up 0.5 μM [3H]putrescine was evaluated in the presence of a variety of structurally related non-radiolabelled compounds, each at a concentration of 50 μM. Results are plotted as a percentage of putrescine uptake obtained without inhibitor (NI). Each value represents the mean ± SD of three independent experiments. E. Abilities of epimastigotes transfected with either pTEX-TcPOT1.1::GFP or pTEX-TcPOT1.2::GFP and grown in putrescine-supplemented SDM-79 medium to take up [14C]cadaverine over a range of cadaverine concentrations. Uptake rates obtained with the control pTEX-TcGRASP::GFP transfectant were subtracted from experimental data points. Data were fitted to a Michaelis-Menten algorithm to determine apparent Km and Vmax values. F. Michaelis-Menten plot of putrescine uptake rates obtained for T. cruzi epimastigotes expressing pTEX-TcPOT1.1::GFP that had been grown either in SDM-79 containing 10% FBS in the absence (○) or presence (●) of 200 μM putrescine for 48 h. The pTEX-TcGRASP::GFP expressing transfectant served as the control for endogenous transport, and control rates were subtracted from the experimental rates. G. Michaelis-Menten analysis of putrescine uptake rates obtained for T. cruzi epimastigotes expressing pTEX-TcPOT1.2::GFP that had been grown either in SDM-79 containing 10% FBS in the absence (○) or presence (●) of 200 μM putrescine for 48 h. The pTEX-GFP expressing transfectant served as the control for endogenous transport and control rates were subtracted from the experimental rates.

Similar articles
-
The Trypanosoma cruzi Diamine Transporter Is Essential for Robust Infection of Mammalian Cells.PLoS One. 2016 Apr 6;11(4):e0152715. doi: 10.1371/journal.pone.0152715. eCollection 2016. PLoS One. 2016. PMID: 27050410 Free PMC article.
-
Structural model of a putrescine-cadaverine permease from Trypanosoma cruzi predicts residues vital for transport and ligand binding.Biochem J. 2013 Jun 15;452(3):423-32. doi: 10.1042/BJ20130350. Biochem J. 2013. PMID: 23535070 Free PMC article.
-
Regulation of a high-affinity diamine transport system in Trypanosoma cruzi epimastigotes.Biochem J. 1996 Jun 1;316 ( Pt 2)(Pt 2):481-6. doi: 10.1042/bj3160481. Biochem J. 1996. PMID: 8687391 Free PMC article.
-
Polyamine metabolism in Trypanosoma cruzi: studies on the expression and regulation of heterologous genes involved in polyamine biosynthesis.Amino Acids. 2010 Feb;38(2):645-51. doi: 10.1007/s00726-009-0425-6. Epub 2009 Dec 3. Amino Acids. 2010. PMID: 19956988 Review.
-
Protean permeases: Diverse roles for membrane transport proteins in kinetoplastid protozoa.Mol Biochem Parasitol. 2019 Jan;227:39-46. doi: 10.1016/j.molbiopara.2018.12.006. Epub 2018 Dec 24. Mol Biochem Parasitol. 2019. PMID: 30590069 Free PMC article. Review.
Cited by
-
The Trypanosoma cruzi Diamine Transporter Is Essential for Robust Infection of Mammalian Cells.PLoS One. 2016 Apr 6;11(4):e0152715. doi: 10.1371/journal.pone.0152715. eCollection 2016. PLoS One. 2016. PMID: 27050410 Free PMC article.
-
Rab11 regulates trafficking of trans-sialidase to the plasma membrane through the contractile vacuole complex of Trypanosoma cruzi.PLoS Pathog. 2014 Jun 26;10(6):e1004224. doi: 10.1371/journal.ppat.1004224. eCollection 2014 Jun. PLoS Pathog. 2014. PMID: 24968013 Free PMC article.
-
TcPho91 is a contractile vacuole phosphate sodium symporter that regulates phosphate and polyphosphate metabolism in Trypanosoma cruzi.Mol Microbiol. 2015 Sep;97(5):911-25. doi: 10.1111/mmi.13075. Epub 2015 Jun 25. Mol Microbiol. 2015. PMID: 26031800 Free PMC article.
-
Cadaverine: a lysine catabolite involved in plant growth and development.Plant Signal Behav. 2013 Oct;8(10):doi: 10.4161/psb.25850. doi: 10.4161/psb.25850. Plant Signal Behav. 2013. PMID: 23887488 Free PMC article.
-
The role of acidocalcisomes in the stress response of Trypanosoma cruzi.Adv Parasitol. 2011;75:307-24. doi: 10.1016/B978-0-12-385863-4.00014-9. Adv Parasitol. 2011. PMID: 21820562 Free PMC article.
References
-
- Algranati ID, Sanchez C, Gonzalez NS. Polyamines in Trypanosoma cruzi and Leishmania mexicana. In: Goldemberg SH, Algranati ID, editors. The Biology and Chemistry of Polyamines. Oxford University Press; Oxford: 1989. pp. 137–146.
-
- Ariyanayagam MR, Fairlamb AH. Diamine auxotrophy may be a universal feature of Trypanosoma cruzi epimastigotes. Mol Biochem Parasitol. 1997;84:111–121. - PubMed
-
- Aronow B, Kaur K, McCartan K, Ullman B. Two high affinity nucleoside transporters in Leishmania donovani. Mol Biochem Parasitol. 1987;22:29–37. - PubMed
-
- Assaraf YG, Golenser J, Spira DT, Messer G, Bachrach U. Cytostatic effect of DL-alpha-difluoromethylornithine against Plasmodium falciparum and its reversal by diamines and spermidine. Parasitol Res. 1987;73:313–318. - PubMed
-
- Bacchi CJ, McCann PP. Parasitic protozoa and polyamines. In: McCann PP, Pegg AE, Sjoerdsma A, editors. Inhibition of Polyamine Metabolism: Biological Significance and Basis for New Therapies. Academic Press; Orlando, FL: 1987. pp. 317–344.
Publication types
MeSH terms
Substances
Associated data
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources

