Triflusal reduces dense-core plaque load, associated axonal alterations and inflammatory changes, and rescues cognition in a transgenic mouse model of Alzheimer's disease
- PMID: 20149872
- PMCID: PMC3707138
- DOI: 10.1016/j.nbd.2010.01.019
Triflusal reduces dense-core plaque load, associated axonal alterations and inflammatory changes, and rescues cognition in a transgenic mouse model of Alzheimer's disease
Abstract
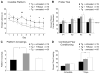
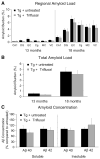
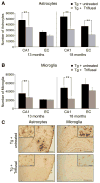
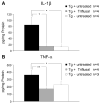
Inflammation has been associated with the two classic lesions in the Alzheimer's (AD) brain, amyloid deposits and neurofibrillary tangles. Recent data suggest that Triflusal, a compound with potent anti-inflammatory effects in the central nervous system in vivo, might delay the conversion from amnestic mild cognitive impairment to a fully established clinical picture of dementia. In the present study, we investigated the effect of Triflusal on brain Abeta accumulation, neuroinflammation, axonal curvature and cognition in an AD transgenic mouse model (Tg2576). Triflusal treatment did not alter the total brain Abeta accumulation but significantly reduced dense-cored plaque load and associated glial cell proliferation, proinflammatory cytokine levels and abnormal axonal curvature, and rescued cognitive deficits in Tg2576 mice. Behavioral benefit was found to involve increased expression of c-fos and BDNF, two of the genes regulated by CREB, as part of the signal transduction cascade underlying the molecular basis of long-term potentiation. These results add preclinical evidence of a potentially beneficial effect of Triflusal in AD.
Figures

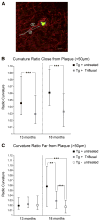
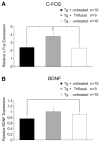
Comment in
-
Thinking outside the box about COX-1 in Alzheimer's disease.Neurobiol Dis. 2010 Jun;38(3):492-4. doi: 10.1016/j.nbd.2010.02.009. Epub 2010 Mar 2. Neurobiol Dis. 2010. PMID: 20206264 Free PMC article.
References
-
- Abel T, Kandel E. Positive and negative regulatory mechanisms that mediate long-term memory storage. Brain Res Rev. 1998;26:360–378. - PubMed
-
- Acarin L, et al. Triflusal posttreatment inhibits glial nuclear factor-kappaB, downregulates the glial response, and is neuroprotective in an excitotoxic injury model in postnatal brain. Stroke. 2001;32:2394–2402. - PubMed
-
- Acarin L, et al. Decrease of proinflammatory molecules correlates with neuroprotective effect of the fluorinated salicylate triflusal after postnatal excitotoxic damage. Stroke. 2002;33:2499–2505. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

