Autoimmunity in membranous nephropathy targets aldose reductase and SOD2
- PMID: 20150532
- PMCID: PMC2831859
- DOI: 10.1681/ASN.2008121259
Autoimmunity in membranous nephropathy targets aldose reductase and SOD2
Abstract
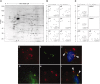
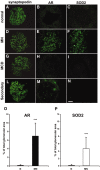
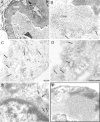
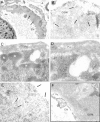
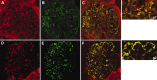
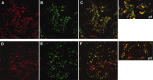
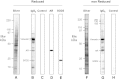
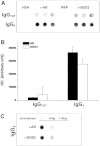
Glomerular targets of autoimmunity in human membranous nephropathy are poorly understood. Here, we used a combined proteomic approach to identify specific antibodies against podocyte proteins in both serum and glomeruli of patients with membranous nephropathy (MN). We detected specific anti-aldose reductase (AR) and anti-manganese superoxide dismutase (SOD2) IgG(4) in sera of patients with MN. We also eluted high titers of anti-AR and anti-SOD2 IgG(4) from microdissected glomeruli of three biopsies of MN kidneys but not from biopsies of other glomerulonephritides characterized by IgG deposition (five lupus nephritis and two membranoproliferative glomerulonephritis). We identified both antigens in MN biopsies but not in other renal pathologies or normal kidney. Confocal and immunoelectron microscopy (IEM) showed co-localization of anti-AR and anti-SOD2 with IgG(4) and C5b-9 in electron-dense podocyte immune deposits. Preliminary in vitro experiments showed an increase of SOD2 expression on podocyte plasma membrane after treatment with hydrogen peroxide. In conclusion, our data support AR and SOD2 as renal antigens of human MN and suggest that oxidative stress may drive glomerular SOD2 expression.
Figures


Similar articles
-
Coexistence of different circulating anti-podocyte antibodies in membranous nephropathy.Clin J Am Soc Nephrol. 2012 Sep;7(9):1394-400. doi: 10.2215/CJN.02170312. Epub 2012 Jul 5. Clin J Am Soc Nephrol. 2012. PMID: 22773590 Free PMC article.
-
Multi-Autoantibody Signature and Clinical Outcome in Membranous Nephropathy.Clin J Am Soc Nephrol. 2020 Dec 7;15(12):1762-1776. doi: 10.2215/CJN.02500220. Epub 2020 Nov 30. Clin J Am Soc Nephrol. 2020. PMID: 33257410 Free PMC article.
-
Idiopathic membranous nephropathy: an autoimmune disease.Semin Nephrol. 2011 Jul;31(4):333-40. doi: 10.1016/j.semnephrol.2011.06.004. Semin Nephrol. 2011. PMID: 21839366 Free PMC article. Review.
-
Direct characterization of target podocyte antigens and auto-antibodies in human membranous glomerulonephritis: Alfa-enolase and borderline antigens.J Proteomics. 2011 Sep 6;74(10):2008-17. doi: 10.1016/j.jprot.2011.05.021. Epub 2011 May 19. J Proteomics. 2011. PMID: 21640210
-
[Secondary membranous nephropathy].Nihon Jinzo Gakkai Shi. 2011;53(5):697-702. Nihon Jinzo Gakkai Shi. 2011. PMID: 21842603 Review. Japanese. No abstract available.
Cited by
-
Changing the concepts of immune-mediated glomerular diseases through proteomics.Proteomics Clin Appl. 2015 Dec;9(11-12):967-71. doi: 10.1002/prca.201400159. Epub 2015 Jun 17. Proteomics Clin Appl. 2015. PMID: 25907758 Free PMC article. Review.
-
Manganese Superoxide Dismutase Dysfunction and the Pathogenesis of Kidney Disease.Front Physiol. 2020 Jul 14;11:755. doi: 10.3389/fphys.2020.00755. eCollection 2020. Front Physiol. 2020. PMID: 32760286 Free PMC article. Review.
-
An autoantigen profile of human A549 lung cells reveals viral and host etiologic molecular attributes of autoimmunity in COVID-19.J Autoimmun. 2021 Jun;120:102644. doi: 10.1016/j.jaut.2021.102644. Epub 2021 Apr 27. J Autoimmun. 2021. PMID: 33971585 Free PMC article.
-
Membranous nephropathy: the start of a paradigm shift.Curr Opin Nephrol Hypertens. 2012 Mar;21(2):203-10. doi: 10.1097/MNH.0b013e32835026ed. Curr Opin Nephrol Hypertens. 2012. PMID: 22240444 Free PMC article. Review.
-
Idiopathic membranous nephropathy and IgG4: an interesting relationship.Clin Nephrol. 2014 Jul;82(1):7-15. doi: 10.5414/CN107768. Clin Nephrol. 2014. PMID: 23380389 Free PMC article. Review.
References
-
- Wasserstein AG: Membranous glomerulonephritis. J Am Soc Nephrol 8: 664–674, 1997 - PubMed
-
- Kerjaschki D: Pathomechanisms and molecular basis of membranous glomerulopathy. Lancet 364: 1194–1196, 2004 - PubMed
-
- Rastaldi MP, Candiano G, Musante L, Bruschi M, Armelloni S, Rimoldi L, Tardanico R, Sanna-Cherchi S, Ferrario F, Montinaro V, Haupt R, Parodi S, Carnevali ML, Allegri L, Camussi G, Gesualdo L, Scolari F, Ghiggeri GM: Glomerular clusterin is associated with PKC-alpha/beta regulation and good outcome of membranous glomerulonephritis in humans. Kidney Int 70: 477–485, 2006 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials

