Hypercapnia causes cellular oxidation and nitrosation in addition to acidosis: implications for CO2 chemoreceptor function and dysfunction
- PMID: 20150563
- PMCID: PMC2886689
- DOI: 10.1152/japplphysiol.01337.2009
Hypercapnia causes cellular oxidation and nitrosation in addition to acidosis: implications for CO2 chemoreceptor function and dysfunction
Abstract
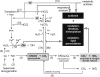
Cellular mechanisms of CO2 chemoreception are discussed and debated in terms of the stimuli produced during hypercapnic acidosis and their molecular targets: protons generated by the hydration of CO2 and dissociation of carbonic acid, which target membrane-bound proteins and lipids in brain stem neurons. The CO2 hydration reaction, however, is not the only reaction that CO2 undergoes that generates molecules capable of modifying proteins and lipids. Molecular CO2 also reacts with peroxynitrite (ONOO-), a reactive nitrogen species (RNS), which is produced from nitric oxide (*NO) and superoxide (*O2-). The CO2/ONOO- reaction, in turn, produces additional nitrosative and oxidative reactive intermediates. Furthermore, protons facilitate additional redox reactions that generate other reactive oxygen species (ROS). ROS/RNS generated by these redox reactions may act as additional stimuli of CO2 chemoreceptors since neurons in chemosensitive areas produce both *NO and *O2- and, therefore, ONOO-. Perturbing *NO, *O2-, and ONOO- activities in chemosensitive areas modulates cardiorespiration. Moreover, neurons in at least one chemosensitive area, the solitary complex, are stimulated by cellular oxidation. Together, these data raise the following two questions: 1) do pH and ROS/RNS work in tandem to stimulate CO2 chemoreceptors during hypercapnic acidosis; and 2) does nitrosative stress and oxidative stress contribute to CO2 chemoreceptor dysfunction? To begin considering these two issues and their implications for central chemoreception, this minireview has the following three goals: 1) summarize the nitrosative and oxidative reactions that occur during hypercapnic acidosis and isocapnic acidosis; 2) review the evidence that redox signaling occurs in chemosensitive areas; and 3) review the evidence that neurons in the solitary complex are stimulated by cellular oxidation.
Figures
References
-
- Aldred AR, Dickson PW, Marley PD, Schreiber G. Distribution of transferrin synthesis in brain and other tissues in the rat. J Biol Chem 262: 5293–5297, 1987 - PubMed
-
- Augusto O, Bonini MG, Amanso AM, Linares E, Santos CC, DeMenezes SL. Nitrogen dioxide and carbonate radical anion: two emerging radicals in biology. Free Radic Biol Med 32: 841–859, 2002 - PubMed
-
- Beckman JS, Koppenol WH. Nitric oxide, superoxide, and peroxynitrite: the good, the bad, and the ugly. Am J Physiol Cell Physiol 271: C1424–C1437, 1996 - PubMed
-
- Bonini MG, Radi R, Ferrer-Sueta G, Ferreira AM, Augusto O. Direct EPR detection of the carbonate radical anion produced from peroxynitrite and carbon dioxide. J Biol Chem 274: 10802–10806, 1999 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

