Adult equine bone marrow stromal cells produce a cartilage-like ECM mechanically superior to animal-matched adult chondrocytes
- PMID: 20153827
- PMCID: PMC2894996
- DOI: 10.1016/j.matbio.2010.02.003
Adult equine bone marrow stromal cells produce a cartilage-like ECM mechanically superior to animal-matched adult chondrocytes
Abstract
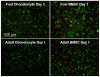
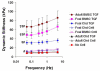
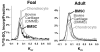
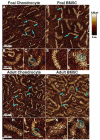
Our objective was to evaluate the age-dependent mechanical phenotype of bone marrow stromal cell- (BMSC-) and chondrocyte-produced cartilage-like neo-tissue and to elucidate the matrix-associated mechanisms which generate this phenotype. Cells from both immature (2-4 month-old foals) and skeletally-mature (2-5 year-old adults) mixed-breed horses were isolated from animal-matched bone marrow and cartilage tissue, encapsulated in self-assembling-peptide hydrogels, and cultured with and without TGF-beta1 supplementation. BMSCs and chondrocytes from both donor ages were encapsulated with high viability. BMSCs from both ages produced neo-tissue with higher mechanical stiffness than that produced by either young or adult chondrocytes. Young, but not adult, chondrocytes proliferated in response to TGF-beta1 while BMSCs from both age groups proliferated with TGF-beta1. Young chondrocytes stimulated by TGF-beta1 accumulated ECM with 10-fold higher sulfated-glycosaminoglycan content than adult chondrocytes and 2-3-fold higher than BMSCs of either age. The opposite trend was observed for hydroxyproline content, with BMSCs accumulating 2-3-fold more than chondrocytes, independent of age. Size-exclusion chromatography of extracted proteoglycans showed that an aggrecan-like peak was the predominant sulfated proteoglycan for all cell types. Direct measurement of aggrecan core protein length and chondroitin sulfate chain length by single molecule atomic force microscopy imaging revealed that, independent of age, BMSCs produced longer core protein and longer chondroitin sulfate chains, and fewer short core protein molecules than chondrocytes, suggesting that the BMSC-produced aggrecan has a phenotype more characteristic of young tissue than chondrocyte-produced aggrecan. Aggrecan ultrastructure, ECM composition, and cellular proliferation combine to suggest a mechanism by which BMSCs produce a superior cartilage-like neo-tissue than either young or adult chondrocytes.
Copyright (c) 2010 Elsevier B.V. All rights reserved.
Figures



Similar articles
-
Adult bone marrow stromal cell-based tissue-engineered aggrecan exhibits ultrastructure and nanomechanical properties superior to native cartilage.Osteoarthritis Cartilage. 2010 Nov;18(11):1477-86. doi: 10.1016/j.joca.2010.07.015. Epub 2010 Aug 6. Osteoarthritis Cartilage. 2010. PMID: 20692354 Free PMC article.
-
Controlled delivery of transforming growth factor β1 by self-assembling peptide hydrogels induces chondrogenesis of bone marrow stromal cells and modulates Smad2/3 signaling.Tissue Eng Part A. 2011 Jan;17(1-2):83-92. doi: 10.1089/ten.TEA.2010.0198. Epub 2010 Sep 22. Tissue Eng Part A. 2011. PMID: 20672992 Free PMC article.
-
Characterization of proteoglycan production and processing by chondrocytes and BMSCs in tissue engineered constructs.Osteoarthritis Cartilage. 2008 Sep;16(9):1092-100. doi: 10.1016/j.joca.2008.01.004. Epub 2008 Feb 21. Osteoarthritis Cartilage. 2008. PMID: 18294870 Free PMC article.
-
Evaluation of adult equine bone marrow- and adipose-derived progenitor cell chondrogenesis in hydrogel cultures.J Orthop Res. 2008 Mar;26(3):322-31. doi: 10.1002/jor.20508. J Orthop Res. 2008. PMID: 17960654
-
[Recent progress of BMSCs acting as seeding cell for tissue engineered cartilage].Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi. 2008 Feb;22(2):163-6. Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi. 2008. PMID: 18365611 Review. Chinese.
Cited by
-
Adult bone marrow stromal cell-based tissue-engineered aggrecan exhibits ultrastructure and nanomechanical properties superior to native cartilage.Osteoarthritis Cartilage. 2010 Nov;18(11):1477-86. doi: 10.1016/j.joca.2010.07.015. Epub 2010 Aug 6. Osteoarthritis Cartilage. 2010. PMID: 20692354 Free PMC article.
-
Effects of Dexamethasone on Mesenchymal Stromal Cell Chondrogenesis and Aggrecanase Activity: Comparison of Agarose and Self-Assembling Peptide Scaffolds.Cartilage. 2013 Jan 1;4(1):63-74. doi: 10.1177/1947603512455196. Cartilage. 2013. PMID: 24533173 Free PMC article.
-
Differences in the intrinsic chondrogenic potential of equine umbilical cord matrix and cord blood mesenchymal stromal/stem cells for cartilage regeneration.Sci Rep. 2018 Sep 14;8(1):13799. doi: 10.1038/s41598-018-28164-9. Sci Rep. 2018. PMID: 30217993 Free PMC article.
-
Hydrogels for the repair of articular cartilage defects.Tissue Eng Part B Rev. 2011 Aug;17(4):281-99. doi: 10.1089/ten.TEB.2011.0077. Epub 2011 Jun 30. Tissue Eng Part B Rev. 2011. PMID: 21510824 Free PMC article. Review.
-
Bioluminescence-mediated longitudinal monitoring of adipose-derived stem cells in a large mammal ex vivo organ culture.Sci Rep. 2015 Sep 9;5:13960. doi: 10.1038/srep13960. Sci Rep. 2015. PMID: 26350622 Free PMC article.
References
-
- Barbero A, Grogan S, Schafer D, Heberer M, Mainil-Varlet P, Martin I. Age related changes in human articular chondrocyte yield, proliferation and post-expansion chondrogenic capacity. Osteoarthritis and Cartilage. 2004;12:476–484. - PubMed
-
- Barry F, Boynton RE, Liu B, Murphy JM. Chondrogenic differentiation of mesenchymal stem cells from bone marrow: differentiation-dependent gene expression of matrix components. Exp Cell Res. 2001;268:189–200. - PubMed
-
- Buckwalter JA, Rosenberg LC. Electron microscopic studies of cartilage proteoglycans. Direct evidence for the variable length of the chondroitin sulfate-rich region of proteoglycan subunit core protein. J Biol Chem. 1982;257:9830–9839. - PubMed
-
- Buckwalter JA, Roughley PJ, Rosenberg LC. Age-related changes in cartilage proteoglycans: quantitative electron microscopic studies. Microsc Res Tech. 1994;28:398–408. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

