Increase in rhamnolipid synthesis under iron-limiting conditions influences surface motility and biofilm formation in Pseudomonas aeruginosa
- PMID: 20154129
- PMCID: PMC2901684
- DOI: 10.1128/JB.01601-09
Increase in rhamnolipid synthesis under iron-limiting conditions influences surface motility and biofilm formation in Pseudomonas aeruginosa
Abstract
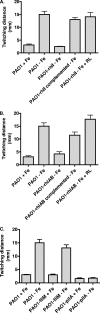
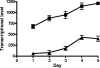
Iron is an essential element for life but also serves as an environmental signal for biofilm development in the opportunistic human pathogen Pseudomonas aeruginosa. Under iron-limiting conditions, P. aeruginosa displays enhanced twitching motility and forms flat unstructured biofilms. In this study, we present evidence suggesting that iron-regulated production of the biosurfactant rhamnolipid is important to facilitate the formation of flat unstructured biofilms. We show that under iron limitation the timing of rhamnolipid expression is shifted to the initial stages of biofilm formation (versus later in biofilm development under iron-replete conditions) and results in increased bacterial surface motility. In support of this observation, an rhlAB mutant defective in biosurfactant production showed less surface motility under iron-restricted conditions and developed structured biofilms similar to those developed by the wild type under iron-replete conditions. These results highlight the importance of biosurfactant production in determining the mature structure of P. aeruginosa biofilms under iron-limiting conditions.
Figures



Similar articles
-
A Survival Strategy for Pseudomonas aeruginosa That Uses Exopolysaccharides To Sequester and Store Iron To Stimulate Psl-Dependent Biofilm Formation.Appl Environ Microbiol. 2016 Oct 14;82(21):6403-6413. doi: 10.1128/AEM.01307-16. Print 2016 Nov 1. Appl Environ Microbiol. 2016. PMID: 27565622 Free PMC article.
-
Influence of quorum sensing and iron on twitching motility and biofilm formation in Pseudomonas aeruginosa.J Bacteriol. 2008 Jan;190(2):662-71. doi: 10.1128/JB.01473-07. Epub 2007 Nov 9. J Bacteriol. 2008. PMID: 17993517 Free PMC article.
-
Coordination of swarming motility, biosurfactant synthesis, and biofilm matrix exopolysaccharide production in Pseudomonas aeruginosa.Appl Environ Microbiol. 2014 Nov;80(21):6724-32. doi: 10.1128/AEM.01237-14. Epub 2014 Aug 29. Appl Environ Microbiol. 2014. PMID: 25172852 Free PMC article.
-
Quorum sensing: implications on rhamnolipid biosurfactant production.Biotechnol Genet Eng Rev. 2010;27:159-84. doi: 10.1080/02648725.2010.10648149. Biotechnol Genet Eng Rev. 2010. PMID: 21415897 Review.
-
Interdependence between iron acquisition and biofilm formation in Pseudomonas aeruginosa.J Microbiol. 2018 Jul;56(7):449-457. doi: 10.1007/s12275-018-8114-3. Epub 2018 Jun 14. J Microbiol. 2018. PMID: 29948830 Free PMC article. Review.
Cited by
-
Microbial siderophores and their potential applications: a review.Environ Sci Pollut Res Int. 2016 Mar;23(5):3984-99. doi: 10.1007/s11356-015-4294-0. Epub 2015 Mar 12. Environ Sci Pollut Res Int. 2016. PMID: 25758420 Review.
-
Efficacy of Artesunate against Pseudomonas aeruginosa Biofilm Mediated by Iron.Biomed Res Int. 2019 Nov 11;2019:4810217. doi: 10.1155/2019/4810217. eCollection 2019. Biomed Res Int. 2019. PMID: 31815139 Free PMC article.
-
Characterization of quorum sensing signals in coral-associated bacteria.Microb Ecol. 2011 May;61(4):783-92. doi: 10.1007/s00248-011-9848-1. Epub 2011 Apr 27. Microb Ecol. 2011. PMID: 21523464
-
Assessing Travel Conditions: Environmental and Host Influences On Bacterial Surface Motility.J Bacteriol. 2018 Mar 19;200(11):e00014-18. doi: 10.1128/JB.00014-18. Online ahead of print. J Bacteriol. 2018. PMID: 29555698 Free PMC article.
-
Why do microorganisms produce rhamnolipids?World J Microbiol Biotechnol. 2012 Feb;28(2):401-19. doi: 10.1007/s11274-011-0854-8. World J Microbiol Biotechnol. 2012. PMID: 22347773 Free PMC article. Review.
References
-
- Berlutti, F., C. Morea, A. Battistoni, S. Sarli, P. Cipriani, F. Superti, M. G. Ammendolia, and P. Valenti. 2005. Iron availability influences aggregation, biofilm, adhesion and invasion of Pseudomonas aeruginosa and Burkholderia cenocepacia. Int. J. Immunopathol. Pharmacol. 18:661-670. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

