Mechanism of error-free and semitargeted mutagenic bypass of an aromatic amine lesion by Y-family polymerase Dpo4
- PMID: 20154704
- PMCID: PMC4215948
- DOI: 10.1038/nsmb.1771
Mechanism of error-free and semitargeted mutagenic bypass of an aromatic amine lesion by Y-family polymerase Dpo4
Abstract
The aromatic amine carcinogen 2-aminofluorene (AF) forms covalent adducts with DNA, predominantly with guanine at the C8 position. Such lesions are bypassed by Y-family polymerases such as Dpo4 via error-free and error-prone mechanisms. We show that Dpo4 catalyzes elongation from a correct 3'-terminal cytosine opposite [AF]G in a nonrepetitive template sequence with low efficiency. This extension leads to cognate full-length product, as well as mis-elongated products containing base mutations and deletions. Crystal structures of the Dpo4 ternary complex, with the 3'-terminal primer cytosine base opposite [AF]G in the anti conformation and with the AF moiety positioned in the major groove, reveal both accurate and misalignment-mediated mutagenic extension pathways. The mutagenic template-primer-dNTP arrangement is promoted by interactions between the polymerase and the bulky lesion rather than by a base pair-stabilized misaligment. Further extension leads to semitargeted mutations via this proposed polymerase-guided mechanism.
Figures
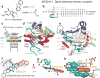


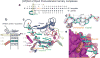



References
-
- Kadlubar FF. DNA adducts of carcinogenic aromatic amines. IARC Sci Publ. 1994;199216 - PubMed
-
- Turesky RJ. Formation and biochemistry of carcinogenic heterocyclic aromatic amines in cooked meats. Toxicol Lett. 2007;168:219–27. - PubMed
-
- Heflich RH, Neft RE. Genetic toxicity of 2-acetylaminofluorene, 2-aminofluorene and some of their metabolites and model metabolites. Mutat Res. 1994;318:73–114. - PubMed
-
- Shibutani S, Suzuki N, Tan X, Johnson F, Grollman AP. Influence of flanking sequence context on the mutagenicity of acetylaminofluorene-derived DNA adducts in mammalian cells. Biochemistry. 2001;40:3717–22. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
- Actions
- Actions
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources

