Toll-like receptor 9 expression in breast and ovarian cancer is associated with poorly differentiated tumors
- PMID: 20156214
- PMCID: PMC3188854
- DOI: 10.1111/j.1349-7006.2010.01491.x
Toll-like receptor 9 expression in breast and ovarian cancer is associated with poorly differentiated tumors
Abstract
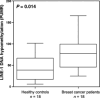
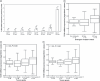
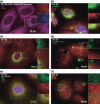
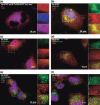
Toll-like receptor 9 (TLR9) activates the innate immune response when exposed to non-methylated CpG-DNA. TLR9 was recently shown to be expressed by cancer cells which have been previously characterized by global hypomethylation. We set out to examine the expression and molecular activity of TLR9 in breast and ovarian cancer cells. Firstly, we confirmed higher levels of hypomethylated DNA in the serum of patients with metastatic breast cancer (n = 18) versus age-matched tumor-free women (n = 18). In breast cancer cell lines and tissues, TLR9 mRNA expression was associated with estrogen-receptor (ER) status (n = 124, P = 0.005). Expression also correlated with increasing tumor grade in both breast (P = 0.03) and ovarian cancer specimens (n = 138, P = 0.04). Immunohistochemical analysis of formalin-fixed paraffin-embedded (FFPE) breast cancer tissues revealed higher TLR9 protein expression in hormone-receptor (HR)-negative specimens (n = 116, P < 0.001). Using an in vitro scratch assay, we observed that cell lines transfected to overexpress TLR9 demonstrated increased cellular migration when stimulated with CpG-DNA. When assessing the molecular activity of TLR9 in breast cancer, we found a strong positive correlation of nuclear factor-kappa B (NF-kappaB) activity with TLR9 mRNA expression (correlation coefficient r = 0.7, P < 0.001). Finally, immunofluorescence analysis of BT-20 and Hs578T breast cancer cell lines showed partial colocalizations of CpG-DNA with TLR9, which diminished when the cells were exposed to methylated CpG-DNA (mCpG-DNA) or control GpC-DNA. In summary we demonstrate that TLR9 expression is associated with poor differentiation in breast and ovarian cancer specimens, and that TLR9 overexpression and stimulation with hypomethylated DNA augments the migratory capacity of cancer cell lines.
Figures


Similar articles
-
Toll-like receptor 9 agonist inhibits ERalpha-mediated transactivation by activating NF-kappaB in breast cancer cell lines.Oncol Rep. 2009 Oct;22(4):935-41. doi: 10.3892/or_00000520. Oncol Rep. 2009. PMID: 19724876
-
Estrogen receptor-α and sex steroid hormones regulate Toll-like receptor-9 expression and invasive function in human breast cancer cells.Breast Cancer Res Treat. 2012 Apr;132(2):411-9. doi: 10.1007/s10549-011-1590-3. Epub 2011 May 24. Breast Cancer Res Treat. 2012. PMID: 21607583
-
Toll-like receptor 9 mediates CpG oligonucleotide-induced cellular invasion.Mol Cancer Res. 2008 Oct;6(10):1534-43. doi: 10.1158/1541-7786.MCR-07-2005. Mol Cancer Res. 2008. PMID: 18922969
-
Toll-like receptor (TLR) gene expression and immunostimulatory effect of CpG oligonucleotides in hormone receptor positive cell line T47D and triple negative breast cancer cell line MDA-MB-468.Immunopharmacol Immunotoxicol. 2020 Oct;42(5):408-415. doi: 10.1080/08923973.2020.1797779. Epub 2020 Aug 6. Immunopharmacol Immunotoxicol. 2020. PMID: 32686546
-
Hypomethylation of the synuclein gamma gene CpG island promotes its aberrant expression in breast carcinoma and ovarian carcinoma.Cancer Res. 2003 Feb 1;63(3):664-73. Cancer Res. 2003. PMID: 12566312
Cited by
-
Polymorphism in Toll-Like Receptors and Helicobacter Pylori Motility in Autoimmune Atrophic Gastritis and Gastric Cancer.Cancers (Basel). 2019 May 10;11(5):648. doi: 10.3390/cancers11050648. Cancers (Basel). 2019. PMID: 31083432 Free PMC article.
-
Hypoxia regulates Toll-like receptor-9 expression and invasive function in human brain cancer cells in vitro.Oncol Lett. 2014 Jul;8(1):266-274. doi: 10.3892/ol.2014.2095. Epub 2014 Apr 25. Oncol Lett. 2014. PMID: 24959259 Free PMC article.
-
Targeting pattern recognition receptors in cancer immunotherapy.Target Oncol. 2012 Mar;7(1):29-54. doi: 10.1007/s11523-012-0213-1. Epub 2012 Mar 8. Target Oncol. 2012. PMID: 22399234 Review.
-
TLR9 Downregulation in Breast Cancer: Its Role in Tumor Immunity, Inflammatory Response, and Cellular Senescence.J Innate Immun. 2025;17(1):354-368. doi: 10.1159/000545527. Epub 2025 Jun 23. J Innate Immun. 2025. PMID: 40550227 Free PMC article.
-
Lower frequency of TLR9 variant associated with protection from breast cancer among African Americans.PLoS One. 2017 Sep 8;12(9):e0183832. doi: 10.1371/journal.pone.0183832. eCollection 2017. PLoS One. 2017. PMID: 28886076 Free PMC article.
References
-
- Takeshita F, Gursel I, Ishii KJ, Suzuki K, Gursel M, Klinman DM. Signal transduction pathways mediated by the interaction of CpG DNA with Toll‐like receptor 9. Semin Immunol 2004; 16: 17–22. - PubMed
-
- Klinman DM. Immunotherapeutic uses of CpG oligodeoxynucleotides. Nat Rev Immunol 2004; 4: 248–57. - PubMed
-
- Fiegl H, Millinger S, Goebel G et al. Circulating tumor‐specific DNA: a marker for monitoring efficacy of adjuvant therapy in cancer patients. Cancer Res 2005; 65: 1141–5. - PubMed
-
- Müller HM, Widschwendter A, Fiegl H et al. DNA methylation in serum of breast cancer patients: An independent prognostic marker. Cancer Res 2003; 63: 7641–5. - PubMed
-
- Jackson K, Yu MC, Arakawa K et al. DNA hypomethylation is prevalent even in low‐grade breast cancers. Cancer Biol Ther 2004; 3: 1225–31. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

