Effect of anti-glycosphingolipid monoclonal antibodies in pathogenic fungal growth and differentiation. Characterization of monoclonal antibody MEST-3 directed to Manpalpha1-->3Manpalpha1-->2IPC
- PMID: 20156351
- PMCID: PMC2831884
- DOI: 10.1186/1471-2180-10-47
Effect of anti-glycosphingolipid monoclonal antibodies in pathogenic fungal growth and differentiation. Characterization of monoclonal antibody MEST-3 directed to Manpalpha1-->3Manpalpha1-->2IPC
Abstract
Background: Studies carried out during the 1990's demonstrated the presence of fungal glycoinositol phosphorylceramides (GIPCs) with unique structures, some of them showed reactivity with sera of patients with histoplasmosis, paracoccidioidomycosis or aspergillosis. It was also observed that fungal GIPCs were able to inhibit T lymphocyte proliferation "in vitro", and studies regarding the importance of these molecules to fungal survival showed that many species of fungi are vulnerable to inhibitors of sphingolipid biosynthesis.
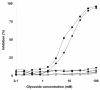
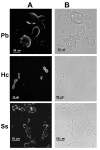
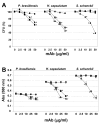
Results: In this paper, we describe a detailed characterization of an IgG2a monoclonal antibody (mAb), termed MEST-3, directed to the Paracoccidioides brasiliensis glycolipid antigen Pb-2 (Manpalpha1-->3Manpalpha1-->2IPC). mAb MEST-3 also recognizes GIPCs bearing the same structure in other fungi. Studies performed on fungal cultures clearly showed the strong inhibitory activity of MEST-3 on differentiation and colony formation of Paracoccidioides brasiliensis, Histoplasma capsulatum and Sporothrix schenckii. Similar inhibitory results were observed when these fungi where incubated with a different mAb, which recognizes GIPCs bearing terminal residues of beta-D-galactofuranose linked to mannose (mAb MEST-1). On the other hand, mAb MEST-2 specifically directed to fungal glucosylceramide (GlcCer) was able to promote only a weak inhibition on fungal differentiation and colony formation.
Conclusions: These results strongly suggest that mAbs directed to specific glycosphingolipids are able to interfere on fungal growth and differentiation. Thus, studies on surface distribution of GIPCs in yeast and mycelium forms of fungi may yield valuable information regarding the relevance of glycosphingolipids in processes of fungal growth, morphological transition and infectivity.
Figures
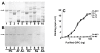



Similar articles
-
Structural diversity and biological significance of glycosphingolipids in pathogenic and opportunistic fungi.Front Cell Infect Microbiol. 2014 Sep 25;4:138. doi: 10.3389/fcimb.2014.00138. eCollection 2014. Front Cell Infect Microbiol. 2014. PMID: 25309884 Free PMC article. Review.
-
Characterization of monoclonal antibody MEST-2 specific to glucosylceramide of fungi and plants.Glycobiology. 2001 Feb;11(2):105-12. doi: 10.1093/glycob/11.2.105. Glycobiology. 2001. PMID: 11287397
-
Structure elucidation of sphingolipids from the mycopathogen Paracoccidioides brasiliensis: an immunodominant beta-galactofuranose residue is carried by a novel glycosylinositol phosphorylceramide antigen.Biochemistry. 1998 Jun 16;37(24):8764-75. doi: 10.1021/bi9730083. Biochemistry. 1998. PMID: 9628738
-
A monoclonal antibody directed to terminal residue of beta-galactofuranose of a glycolipid antigen isolated from Paracoccidioides brasiliensis: cross-reactivity with Leishmania major and Trypanosoma cruzi.Glycobiology. 1997 Jun;7(4):463-8. doi: 10.1093/glycob/7.4.463. Glycobiology. 1997. PMID: 9184826
-
Dimorphism in pathogenic fungi.Crit Rev Microbiol. 1980;8(1):49-97. doi: 10.3109/10408418009085078. Crit Rev Microbiol. 1980. PMID: 6996918 Review. No abstract available.
Cited by
-
Paracoccidioidomycosis Protective Immunity.J Fungi (Basel). 2021 Feb 13;7(2):137. doi: 10.3390/jof7020137. J Fungi (Basel). 2021. PMID: 33668671 Free PMC article. Review.
-
Galf-Specific Neolectins: Towards Promising Diagnostic Tools.Int J Mol Sci. 2024 Apr 28;25(9):4826. doi: 10.3390/ijms25094826. Int J Mol Sci. 2024. PMID: 38732045 Free PMC article.
-
Structural diversity and biological significance of glycosphingolipids in pathogenic and opportunistic fungi.Front Cell Infect Microbiol. 2014 Sep 25;4:138. doi: 10.3389/fcimb.2014.00138. eCollection 2014. Front Cell Infect Microbiol. 2014. PMID: 25309884 Free PMC article. Review.
-
Antibodies Against Glycolipids Enhance Antifungal Activity of Macrophages and Reduce Fungal Burden After Infection with Paracoccidioides brasiliensis.Front Microbiol. 2016 Feb 3;7:74. doi: 10.3389/fmicb.2016.00074. eCollection 2016. Front Microbiol. 2016. PMID: 26870028 Free PMC article.
-
Antifungal Drug Development: Targeting the Fungal Sphingolipid Pathway.J Fungi (Basel). 2020 Aug 20;6(3):142. doi: 10.3390/jof6030142. J Fungi (Basel). 2020. PMID: 32825250 Free PMC article. Review.
References
-
- Drouhet E. In: Medical Mycology. Ajello L, Hay R, editor. Arnold New York; 1998. Historical introduction; pp. 3–42.
-
- Takesako K, Kuroda H, Inoue T, Haruna F, Yoshikawa Y, Kato I, Uchida K, Hiratani T, Yamaguchi H. Biological properties of aureobasidin A, a cyclic depsipeptide antifungal antibiotic. J Antibiot. 1993;46:1414–1420. - PubMed
-
- Nagiec MM, Nagiec EE, Baltisberger JA, Wells GB, Lester RL, Dickson RC. Sphingolipid synthesis as a target for antifungal drugs. Complementation of the inositol phosphorylceramide synthase defect in a mutant strain of Saccharomyces cerevisiae by the AUR1 gene. J Biol Chem. 1997;272:9809–9817. doi: 10.1074/jbc.272.15.9809. - DOI - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

