Randomized phase III clinical trial of five different arms of treatment in 332 patients with cancer cachexia
- PMID: 20156909
- PMCID: PMC3227938
- DOI: 10.1634/theoncologist.2009-0153
Randomized phase III clinical trial of five different arms of treatment in 332 patients with cancer cachexia
Abstract
Purpose: A phase III, randomized study was carried out to establish the most effective and safest treatment to improve the primary endpoints of cancer cachexia-lean body mass (LBM), resting energy expenditure (REE), and fatigue-and relevant secondary endpoints: appetite, quality of life, grip strength, Glasgow Prognostic Score (GPS) and proinflammatory cytokines.
Patients and methods: Three hundred thirty-two assessable patients with cancer-related anorexia/cachexia syndrome were randomly assigned to one of five treatment arms: arm 1, medroxyprogesterone (500 mg/day) or megestrol acetate (320 mg/day); arm 2, oral supplementation with eicosapentaenoic acid; arm 3, L-carnitine (4 g/day); arm 4, thalidomide (200 mg/day); and arm 5, a combination of the above. Treatment duration was 4 months.
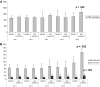
Results: Analysis of variance showed a significant difference between treatment arms. A post hoc analysis showed the superiority of arm 5 over the others for all primary endpoints. An analysis of changes from baseline showed that LBM (by dual-energy X-ray absorptiometry and by L3 computed tomography) significantly increased in arm 5. REE decreased significantly and fatigue improved significantly in arm 5. Appetite increased significantly in arm 5; interleukin (IL)-6 decreased significantly in arm 5 and arm 4; GPS and Eastern Cooperative Oncology Group performance status (ECOG PS) score decreased significantly in arm 5, arm 4, and arm 3. Toxicity was quite negligible, and was comparable between arms.
Conclusion: The most effective treatment in terms of all three primary efficacy endpoints and the secondary endpoints appetite, IL-6, GPS, and ECOG PS score was the combination regimen that included all selected agents.
Conflict of interest statement
The content of this article has been reviewed by independent peer reviewers to ensure that it is balanced, objective, and free from commercial bias. No financial relationships relevant to the content of this article have been disclosed by the authors or independent peer reviewers.
Figures
Comment in
-
More is better: a multimodality approach to cancer cachexia.Oncologist. 2010;15(2):119-21. doi: 10.1634/theoncologist.2010-0019. Epub 2010 Feb 4. Oncologist. 2010. PMID: 20133501 Free PMC article.
References
-
- Khan ZH, Simpson EJ, Cole AT, et al. Oesophageal cancer and cachexia: The effect of short-term treatment with thalidomide on weight loss and lean body mass. Aliment Pharmacol Ther. 2003;17:677–682. - PubMed
-
- Heber D, Byerley LO, Chi J, et al. Pathophysiology of malnutrition in the adult cancer patient. Cancer. 1986;58(8 suppl):1867–1873. - PubMed
-
- Bruera E. Clinical management of anorexia and cachexia in patients with advanced cancer. Oncology. 1992;49(suppl 2):35–42. - PubMed
-
- Brennan MF. Uncomplicated starvation versus cancer cachexia. Cancer Res. 1977;37:2359–2364. - PubMed
-
- Nelson K, Walsh D. Management of the anorexia/cachexia syndrome. Cancer Bull. 1991;43:403–406.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

