Coronary heart disease in postmenopausal recipients of estrogen plus progestin therapy: does the increased risk ever disappear? A randomized trial
- PMID: 20157135
- PMCID: PMC2936769
- DOI: 10.7326/0003-4819-152-4-201002160-00005
Coronary heart disease in postmenopausal recipients of estrogen plus progestin therapy: does the increased risk ever disappear? A randomized trial
Abstract
Background: Estrogen plus progestin therapy increases the risk for coronary heart disease (CHD) in postmenopausal women. However, this increased risk might be limited to the first years of use and to women who start therapy late in menopause.
Objective: To estimate the effect of continuous estrogen plus progestin therapy on CHD risk over time and stratified by years since menopause.
Design: Women's Health Initiative randomized, double-blinded, placebo-controlled trial. (ClinicalTrials.gov registration number: NCT00000611)
Setting: 40 U.S. clinical centers.
Patients: 16 608 postmenopausal women with an intact uterus at baseline from 1993 to 1998.
Intervention: Conjugated equine estrogens, 0.625 mg/d, plus medroxyprogesterone acetate, 2.5 mg/d, or placebo.
Measurements: Adherence-adjusted hazard ratios and CHD-free survival curves estimated through inverse probability weighting.
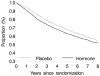
Results: Compared with no use of hormone therapy, the hazard ratio for continuous use of estrogen plus progestin therapy was 2.36 (95% CI, 1.55 to 3.62) for the first 2 years and 1.69 (CI, 0.98 to 2.89) for the first 8 years. For women within 10 years after menopause, the hazard ratios were 1.29 (CI, 0.52 to 3.18) for the first 2 years and 0.64 (CI, 0.21 to 1.99) for the first 8 years, and the CHD-free survival curves for continuous use and no use of estrogen plus progestin crossed at about 6 years (CI, 2 years to 10 years).
Limitation: The analysis may not have fully adjusted for joint determinants of adherence and CHD risk. Sample sizes for some subgroup analyses were small.
Conclusion: No suggestion of a decreased risk for CHD was found within the first 2 years of estrogen plus progestin use, including in women who initiated therapy within 10 years after menopause. A possible cardioprotective effect in these women who initiated therapy closer to menopause became apparent only after 6 years of use.
Primary funding source: National Heart, Lung, and Blood Institute.
Figures


Comment in
-
Combined estrogen plus progestin may reduce coronary heart disease risk after 5 years of use in postmenopausal women starting treatment within 10 years of menopause.Evid Based Med. 2010 Jun;15(3):69-70. doi: 10.1136/ebm1079. Evid Based Med. 2010. PMID: 20522676 No abstract available.
-
Menopausal hormone therapy and risk for cardiovascular disease in the WHI trial.Ann Intern Med. 2010 Jul 6;153(1):60-1; author reply 61-2. doi: 10.7326/0003-4819-153-1-201007060-00019. Ann Intern Med. 2010. PMID: 20621909 No abstract available.
-
Menopausal hormone therapy and risk for cardiovascular disease in the WHI trial.Ann Intern Med. 2010 Jul 6;153(1):61; author reply 61-2. doi: 10.7326/0003-4819-153-1-201007060-00021. Ann Intern Med. 2010. PMID: 20621911 No abstract available.
-
Menopausal hormone therapy and risk for cardiovascular disease in the WHI trial.Ann Intern Med. 2010 Jul 6;153(1):61; author reply 61-2. doi: 10.7326/0003-4819-153-1-201007060-00020. Ann Intern Med. 2010. PMID: 20621912 No abstract available.
Summary for patients in
-
Summaries for patients: The effect of estrogen plus progestin on coronary heart disease.Ann Intern Med. 2010 Feb 16;152(4):I-40. doi: 10.7326/0003-4819-152-4-201002160-00002. Ann Intern Med. 2010. PMID: 20157120 No abstract available.
References
-
- Manson JE, Hsia J, Johnson KC, Rossouw JE, Assaf AR, Lasser NL, et al. Estrogen plus progestin and the risk of coronary heart disease. N Engl J Med. 2003;349(6):523–34. - PubMed
-
- Hernán MA, Robins JM, García Rodríguez LA. Discussion of Statistical issues arising in the Women's Health Initiative by Prentice RL, Pettinger M, Andreson GL. Biometrics. 2005;61(4):922–930. - PubMed
-
- Manson JE, Bassuk SS. Invited commentary: hormone therapy and risk of coronary heart disease why renew the focus on the early years of menopause? Am J Epidemiol. 2007;166(5):511–7. - PubMed
-
- Mendelsohn ME, Karas RH. HRT and the young at heart. N Engl J Med. 2007;356(25):2639–41. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
