A Comparison of Cardiovascular Biomarkers in Patients Treated for Three Months with Etoricoxib, Celecoxib, Ibuprofen, and Placebo
- PMID: 20157362
- PMCID: PMC2817438
- DOI: 10.1111/j.1753-5174.2007.00002.x
A Comparison of Cardiovascular Biomarkers in Patients Treated for Three Months with Etoricoxib, Celecoxib, Ibuprofen, and Placebo
Abstract
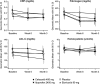
OBJECTIVES: Selective cyclooxygenase (COX)-2 inhibitors are effective analgesic and anti-inflammatory agents with improved gastrointestinal safety and tolerability compared with traditional NSAIDs. However, data from long-term, placebo-controlled studies have shown an increased risk of thrombotic cardiovascular (CV) events for COX-2 inhibitors. Changes in levels of CV biomarkers are potentially useful surrogate measures of pathologic changes associated with CV risk. METHODS: We randomized 433 patients with osteoarthritis to etoricoxib 90 mg once daily, celecoxib 200 mg twice daily, ibuprofen 800 mg three times daily, or placebo for 12 weeks. The hypothesis was that etoricoxib would be non-inferior or superior to placebo in effect on C-reactive protein (CRP), LDL-cholesterol, homocysteine, and fibrinogen. RESULTS: Relative to placebo, etoricoxib was noninferior for effect on CRP (decreased 7.8% vs. placebo; 97.5% CI of the difference: -30.5, 22.4), LDL-C (-4.0% vs. placebo; 97.5% CI: -10.6, 3.2), homocysteine (-3.9% vs. placebo; 97.5% CI: -11.6, 4.6), and fibrinogen (-3.7% vs. placebo; 97.5% CI: -9.4, 2.3). Etoricoxib was not different from placebo, celecoxib, or ibuprofen for any biomarker. CONCLUSION: Etoricoxib was comparable to placebo, celecoxib, and ibuprofen for effects on the CV risk markers measured.
Figures

References
-
- Bresalier RS, Sandler RS, Quan H, et al. Cardiovascular events associated with rofecoxib in a colorectal adenoma chemoprevention trial. N Engl J Med. 2005;352:1092–102. - PubMed
-
- Solomon SD, McMurray JJ, Pfeffer MA, et al. Cardiovascular risk associated with celecoxib in a clinical trial for colorectal adenoma prevention. N Engl J Med. 2005;352:1071–80. - PubMed
-
- FitzGerald GA. Coxibs Cardiovascular Disease. N Engl J Med. 2004;351:2875–8. - PubMed
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous
