Cyclooxygenase-1 null mice show reduced neuroinflammation in response to beta-amyloid
- PMID: 20157512
- PMCID: PMC2806008
- DOI: 10.18632/aging.100021
Cyclooxygenase-1 null mice show reduced neuroinflammation in response to beta-amyloid
Abstract
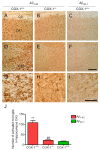
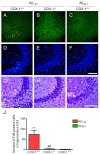
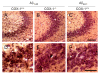
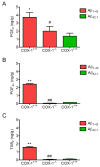
Several independent epidemiological studies indicate that chronic use of non-steroidal anti-inflammatory drugs can reduce the risk of developing Alzheimer's disease (AD), supporting the inflammatory cascade hypothesis. Although the first clinical trial with indomethacin, a preferential cyclooxygenase (COX)-1 inhibitor, showed beneficial effects, subsequent large clinical trials, mostly using COX-2 inhibitors, failed to show any beneficial effect in AD patients with mild to severe cognitive impairment. These combined data suggest that either an early treatment is crucial to stop the mechanisms underlying the disease before the onset of the symptoms, or that preferential COX-1 inhibition, rather than COX-2, is beneficial. Therefore, a full understanding of the physiological, pathological, and/or neuroprotective role of COX isoforms may help to develop better therapeutic strategies for the prevention or treatment of AD. In this study, we examined the effect of COX-1 genetic deletion on the inflammatory response and neurodegeneration induced by beta-amyloid. beta-amyloid (Abeta(1-42)) was centrally injected in the lateral ventricle of COX-1-deficient (COX-1(-/-)) and their respective wild-type (WT) mice. In COX-1(-/-) mice, Abeta(1-42)-induced inflammatory response and neuronal damage were attenuated compared to WT mice, as shown by Fluoro-Jade B and nitrotyrosine staining. These results indicate that inhibition of COX-1 activity may be valid therapeutic strategy to reduce brain inflammatory response and neurodegeneration.
Keywords: cyclooxygenase; microglia; neuroinflammation; neurotoxicity; oxidative stress.
Conflict of interest statement
The authors in this manuscript have no conflict of interests to declare.
Figures

Comment in
-
A role for cyclooxygenase-1 in beta-amyloid-induced neuroinflammation.Aging (Albany NY). 2009 Apr 13;1(4):350-3. doi: 10.18632/aging.100039. Aging (Albany NY). 2009. PMID: 20157521 Free PMC article. No abstract available.
Similar articles
-
Hepcidin attenuates amyloid beta-induced inflammatory and pro-oxidant responses in astrocytes and microglia.J Neurochem. 2017 Jul;142(1):140-152. doi: 10.1111/jnc.14005. Epub 2017 Apr 4. J Neurochem. 2017. PMID: 28266714
-
Detection of Cyclooxygenase-1 in Activated Microglia During Amyloid Plaque Progression: PET Studies in Alzheimer's Disease Model Mice.J Nucl Med. 2016 Feb;57(2):291-6. doi: 10.2967/jnumed.115.166116. Epub 2015 Nov 19. J Nucl Med. 2016. PMID: 26585055
-
Adiponectin suppresses amyloid-β oligomer (AβO)-induced inflammatory response of microglia via AdipoR1-AMPK-NF-κB signaling pathway.J Neuroinflammation. 2019 May 25;16(1):110. doi: 10.1186/s12974-019-1492-6. J Neuroinflammation. 2019. PMID: 31128596 Free PMC article.
-
Cyclooxygenase-1 and -2 in the different stages of Alzheimer's disease pathology.Curr Pharm Des. 2008;14(14):1419-27. doi: 10.2174/138161208784480171. Curr Pharm Des. 2008. PMID: 18537664 Review.
-
Amyloid beta-peptide (1-42)-induced oxidative stress and neurotoxicity: implications for neurodegeneration in Alzheimer's disease brain. A review.Free Radic Res. 2002 Dec;36(12):1307-13. doi: 10.1080/1071576021000049890. Free Radic Res. 2002. PMID: 12607822 Review.
Cited by
-
Thiamine Compounds Alleviate Oxidative Stress, Over-Expression of Pro-Inflammatory Markers and Behavioral Abnormalities in a Mouse Predation Model of PTSD.Int J Mol Sci. 2025 Jul 10;26(14):6627. doi: 10.3390/ijms26146627. Int J Mol Sci. 2025. PMID: 40724875 Free PMC article.
-
Cyclooxygenase-1 deletion in 5 × FAD mice protects against microglia-induced neuroinflammation and mitigates cognitive impairment.Transl Neurodegener. 2025 Aug 22;14(1):43. doi: 10.1186/s40035-025-00501-9. Transl Neurodegener. 2025. PMID: 40847374 Free PMC article.
-
Cerium Oxide Nanoparticles Protect against Oxidant Injury and Interfere with Oxidative Mediated Kinase Signaling in Human-Derived Hepatocytes.Int J Mol Sci. 2019 Nov 27;20(23):5959. doi: 10.3390/ijms20235959. Int J Mol Sci. 2019. PMID: 31783479 Free PMC article.
-
Prostacyclin Promotes Degenerative Pathology in a Model of Alzheimer's Disease.Front Cell Neurosci. 2022 Feb 7;16:769347. doi: 10.3389/fncel.2022.769347. eCollection 2022. Front Cell Neurosci. 2022. PMID: 35197825 Free PMC article.
-
Cyclooxygenases-1 and -2 differentially modulate leukocyte recruitment into the inflamed brain.Pharmacogenomics J. 2010 Oct;10(5):448-57. doi: 10.1038/tpj.2009.68. Epub 2009 Dec 29. Pharmacogenomics J. 2010. PMID: 20038958 Free PMC article.
References
-
- Yermakova AV, Rollins J, Callahan LM, Rogers J, O'Banion MK. Cyclooxygenase-1 in human Alzheimer and control brain: quantitative analysis of expression by microglia and CA3 hippocampal neurons. J Neuropathol Exp Neurol. 1999;58:1135–1146. - PubMed
-
- Ho L, Luterman JD, Aisen PS, Pasinetti GM, Montine TJ, Morrow JD. Elevated CSF prostaglandin E2 levels in patients with probable AD. Neurology. 2000;55:323. - PubMed
-
- Hoozemans JJ, Rozemuller AJ, Janssen I, De Groot CJ, Veerhuis R, Eikelenboom P. Cyclooxygenase expression in microglia and neurons in Alzheimer's disease and control brain. Acta Neuropathol (Berl) 2001;101:2–8. - PubMed
-
- Minghetti L. Cyclooxygenase-2 (COX-2) in inflammatory and degenerative brain diseases. J Neuropathol Exp Neurol. 2004;63:901–910. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
