Self-assembling multidomain peptide hydrogels: designed susceptibility to enzymatic cleavage allows enhanced cell migration and spreading
- PMID: 20158218
- PMCID: PMC2857986
- DOI: 10.1021/ja910481t
Self-assembling multidomain peptide hydrogels: designed susceptibility to enzymatic cleavage allows enhanced cell migration and spreading
Abstract
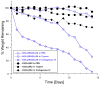
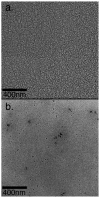
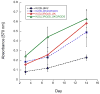
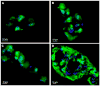
Multidomain peptides are a class of amphiphilic self-assembling peptides with a modular ABA block motif in which the amphiphilic B block drives self-assembly while the flanking A blocks, which are electrostatically charged, control the conditions under which assembly takes place. Previously we have shown that careful selection of the amino acids in the A and B blocks allow one to control the self-assembled fiber length and viscoelastic properties of formed hydrogels. Here we demonstrate how the modular nature of this peptide assembler can be designed for biological applications. With control over fiber length and diameter, gelation conditions, and viscoelastic properties, we can develop suitable materials for biological applications. Going beyond a simple carrier for cell delivery, a biofunctional scaffold will interact with the cells it carries, promoting advantageous cell-matrix interactions. We demonstrate the design of a multidomain peptide into a bioactive variant by incorporation of a matrix metalloprotease 2 (MMP-2) specific cleavage site and cell adhesion motif. Gel formation and rheological properties were assessed and compared to related peptide hydrogels. Proteolytic degradation by collagenase IV was observed in a gel weight loss study and confirmed by specific MMP-2 degradation monitored by mass spectrometry and cryo-transmission electron microscopy (cryo-TEM). Combination of this cleavage site with the cell adhesion motif RGD resulted in increased cell viability and cell spreading and encouraged cell migration into the hydrogel matrix. Collectively the structural, mechanical, and bioactive properties of this multidomain peptide hydrogel make it suitable as an injectable material for a variety of tissue engineering applications.
Figures


Similar articles
-
Self-assembled peptide-based hydrogels as scaffolds for anchorage-dependent cells.Biomaterials. 2009 May;30(13):2523-30. doi: 10.1016/j.biomaterials.2009.01.010. Epub 2009 Feb 7. Biomaterials. 2009. PMID: 19201459
-
Synthesis and characterization of matrix metalloprotease sensitive-low molecular weight hyaluronic acid based hydrogels.J Mater Sci Mater Med. 2008 Nov;19(11):3311-8. doi: 10.1007/s10856-008-3469-3. Epub 2008 May 22. J Mater Sci Mater Med. 2008. PMID: 18496734
-
Functionalized self-assembling peptide nanofiber hydrogels mimic stem cell niche to control human adipose stem cell behavior in vitro.Acta Biomater. 2013 Jun;9(6):6798-805. doi: 10.1016/j.actbio.2013.01.027. Epub 2013 Feb 4. Acta Biomater. 2013. PMID: 23380207
-
Biomimetic Self-Assembling Peptide Hydrogels for Tissue Engineering Applications.Adv Exp Med Biol. 2018;1064:297-312. doi: 10.1007/978-981-13-0445-3_18. Adv Exp Med Biol. 2018. PMID: 30471040 Review.
-
Self-assembly of short peptides to form hydrogels: design of building blocks, physical properties and technological applications.Acta Biomater. 2014 Apr;10(4):1671-82. doi: 10.1016/j.actbio.2013.08.013. Epub 2013 Aug 16. Acta Biomater. 2014. PMID: 23958781 Review.
Cited by
-
Current Advance and Future Prospects of Tissue Engineering Approach to Dentin/Pulp Regenerative Therapy.Stem Cells Int. 2016;2016:9204574. doi: 10.1155/2016/9204574. Epub 2016 Mar 16. Stem Cells Int. 2016. PMID: 27069484 Free PMC article. Review.
-
Self-assembling Molecular Medicine for the Subacute Phase of Ischemic Stroke.Neurochem Res. 2022 Sep;47(9):2488-2498. doi: 10.1007/s11064-022-03638-5. Epub 2022 Jun 6. Neurochem Res. 2022. PMID: 35666393 Free PMC article. Review.
-
Thiol-Ene Photo-Click Collagen-PEG Hydrogels: Impact of Water-Soluble Photoinitiators on Cell Viability, Gelation Kinetics and Rheological Properties.Polymers (Basel). 2017 Jun 14;9(6):226. doi: 10.3390/polym9060226. Polymers (Basel). 2017. PMID: 30970903 Free PMC article.
-
Evolving the use of peptides as components of biomaterials.Biomaterials. 2011 Jun;32(18):4198-204. doi: 10.1016/j.biomaterials.2011.02.030. Biomaterials. 2011. PMID: 21515167 Free PMC article.
-
Multidomain Peptide Hydrogel Accelerates Healing of Full-Thickness Wounds in Diabetic Mice.ACS Biomater Sci Eng. 2018;4(4):1386-1396. doi: 10.1021/acsbiomaterials.8b00031. Epub 2018 Mar 16. ACS Biomater Sci Eng. 2018. PMID: 29687080 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

