Primary changes of the mechanical properties of Southern Bean Mosaic Virus upon calcium removal
- PMID: 20159165
- PMCID: PMC2820651
- DOI: 10.1016/j.bpj.2009.10.047
Primary changes of the mechanical properties of Southern Bean Mosaic Virus upon calcium removal
Abstract
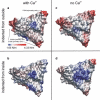
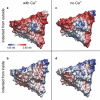
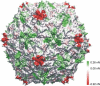
The mechanical properties of viral shells are crucial determinates for the pathway and mechanism by which the genetic material leaves the capsid during infection and have therefore been studied by atomic force microscopy as well as by atomistic simulations. The mechanical response to forces from inside the capsid are found to be relevant, especially after ion removal from the shell structure, which is generally assumed to be essential during viral infection; however, atomic force microscopy measurements are restricted to probing the capsids from outside, and the primary effect of ion removal is still inaccessible. To bridge this gap, we performed atomistic force-probe molecular dynamics simulations of the complete solvated icosahedral shell of Southern Bean Mosaic Virus and compared the distribution of elastic constants and yielding forces on the icosahedral shell for probing from inside with the distribution of outside mechanical properties obtained previously. Further, the primary effect of calcium removal on the mechanical properties on both sides, as well as on their spatial distribution, is quantified. Marked differences are seen particularly at the pentamer centers, although only small structural changes occur on the short timescales of the simulation. This unexpected primary effect, hence, precedes subsequent effects due to capsid swelling. In particular, assuming that genome release is preceded by an opening of capsomers instead of a complete capsid bursting, our observed weakening along the fivefold symmetry axes let us suggest pentamers as possible exit ports for RNA release.
Copyright 2010 Biophysical Society. Published by Elsevier Inc. All rights reserved.
Figures

Similar articles
-
Mechanical properties of the icosahedral shell of southern bean mosaic virus: a molecular dynamics study.Biophys J. 2009 Feb 18;96(4):1350-63. doi: 10.1016/j.bpj.2008.11.028. Biophys J. 2009. PMID: 19217853 Free PMC article.
-
Structure of sesbania mosaic virus at 3 A resolution.Structure. 1995 Oct 15;3(10):1021-30. doi: 10.1016/s0969-2126(01)00238-6. Structure. 1995. PMID: 8589997
-
Role of metal ion-mediated interactions in the assembly and stability of Sesbania mosaic virus T=3 and T=1 capsids.J Mol Biol. 2004 Sep 17;342(3):1001-14. doi: 10.1016/j.jmb.2004.07.022. J Mol Biol. 2004. PMID: 15342252
-
Constraints on the assembly of spherical virus particles.Virology. 1984 Apr 15;134(1):1-11. doi: 10.1016/0042-6822(84)90267-8. Virology. 1984. PMID: 6710869
-
Structure of Sesbania mosaic virus at 3 A resolution.Biophys Chem. 1997 Oct;68(1-3):33-42. doi: 10.1016/s0301-4622(97)00005-7. Biophys Chem. 1997. PMID: 9468608
Cited by
-
Computational virology: From the inside out.Biochim Biophys Acta. 2016 Jul;1858(7 Pt B):1610-8. doi: 10.1016/j.bbamem.2016.02.007. Epub 2016 Feb 10. Biochim Biophys Acta. 2016. PMID: 26874202 Free PMC article. Review.
-
Swelling and softening of the cowpea chlorotic mottle virus in response to pH shifts.Biophys J. 2015 May 19;108(10):2541-2549. doi: 10.1016/j.bpj.2015.04.019. Biophys J. 2015. PMID: 25992732 Free PMC article.
-
Minimal Cylinder Analysis Reveals the Mechanical Properties of Oncogenic Nucleosomes.Biophys J. 2020 May 5;118(9):2309-2318. doi: 10.1016/j.bpj.2020.01.042. Epub 2020 Feb 12. Biophys J. 2020. PMID: 32097625 Free PMC article.
-
Overview on Sobemoviruses and a Proposal for the Creation of the Family Sobemoviridae.Viruses. 2015 Jun 15;7(6):3076-115. doi: 10.3390/v7062761. Viruses. 2015. PMID: 26083319 Free PMC article. Review.
-
Elastic properties and heterogeneous stiffness of the phi29 motor connector channel.Biophys J. 2014 Mar 18;106(6):1338-48. doi: 10.1016/j.bpj.2014.01.028. Biophys J. 2014. PMID: 24655509 Free PMC article.
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

