Evaluating oral noncombustible potential-reduced exposure products for smokers
- PMID: 20159791
- PMCID: PMC2847072
- DOI: 10.1093/ntr/ntq003
Evaluating oral noncombustible potential-reduced exposure products for smokers
Abstract
Introduction: Potential-reduced exposure products (PREPs) are marketed as a way for smokers to continue using tobacco while possibly lessening their tobacco toxicant intake. Some tobacco-based PREPs are combustible and intended to be smoked, while others are noncombustible and intended to be administered orally (e.g., Camel Snus [CS] tobacco sachets and Ariva tobacco tablets). The ability of these noncombustible PREPs to reduce smokers' exposure to cigarette-delivered toxicants and suppress tobacco abstinence symptoms effectively is unclear. Clinical laboratory methods have been used to measure combustible PREP-associated toxicant exposure and abstinence symptom suppression and could be applied to evaluating the effects of orally administered noncombustible PREPs.
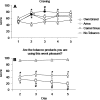
Methods: In this study, 21 smokers (6 women) participated in four 5-day conditions that differed by product used: CS, Ariva, own brand cigarettes, or no tobacco. Measures included expired-air carbon monoxide (CO), the urinary metabolite of nicotine (cotinine), the urinary metabolite of the carcinogen NNK (NNAL-T), and subjective effect ratings.
Results: Relative to own brand, all other conditions were associated with CO and cotinine levels that were lower and abstinence symptom ratings that were greater. Only no-tobacco use was associated with significantly lower NNAL levels. Acceptability ratings were also lower in all conditions relative to own brand.
Discussion: Although these oral products reduce exposure to CO, their ineffective abstinence symptom suppression and low acceptability may limit their viability as PREPs. As with combustible PREPs, clinical laboratory study of orally administered noncombustible PREPs will be a valuable part of any comprehensive PREP evaluation strategy.
Figures

References
-
- Acosta MC, Buchhalter AR, Breland AB, Hamilton DCP, Eissenberg T. Urine cotinine as an index of smoking status in abstinent smokers: Comparison of GC/MS with immunoassay test strip. Nicotine & Tobacco Research. 2004;6:615–620. - PubMed
-
- Bolliger CT, Zellweger JP, Danielsson T, van Biljon X, Robidou A, Westin A, et al. Influence of long-term smoking reduction on health risk markers and quality of life. Nicotine & Tobacco Research. 2002;4:433–439. - PubMed
-
- Breland AB, Buchhalter AR, Evans SE, Eissenberg T. Evaluating acute effects of potential reduced-exposure products for smokers: Clinical laboratory methodology. Nicotine & Tobacco Research. 2002;4:S131–S140. - PubMed
-
- Breland AB, Kleykamp BA, Eissenberg T. Clinical laboratory evaluation of potential reduced exposure products for smokers. Nicotine & Tobacco Research. 2006;8:727–738. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

