Cholesterol Supplementation During Production Increases the Infectivity of Retroviral and Lentiviral Vectors Pseudotyped with the Vesicular Stomatitis Virus Glycoprotein (VSV-G)
- PMID: 20160854
- PMCID: PMC2663912
- DOI: 10.1016/j.bej.2008.12.004
Cholesterol Supplementation During Production Increases the Infectivity of Retroviral and Lentiviral Vectors Pseudotyped with the Vesicular Stomatitis Virus Glycoprotein (VSV-G)
Abstract
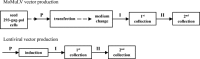
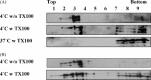
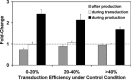
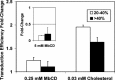
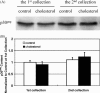
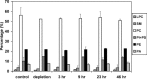
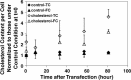
Cholesterol, a major component of plasma membrane lipid rafts, is important for assembly and budding of enveloped viruses, including influenza and HIV-1. Cholesterol depletion impairs virus assembly and infectivity. This study examined the effects of exogenous cholesterol addition (delivered as a complex with methyl beta cyclodextrin) on the production of Molony murine leukemia virus retroviral vector and HIV-1-based lentiviral vector pseudotyped with the vesicular stomatitis virus glycoprotein (VSV-G). Cholesterol supplementation before and during vector production enhanced the infectivity of retroviral and lentiviral vectors up to 4-fold and 6-fold, respectively. In contrast, the amount of retroviral vector produced was unchanged, and that of lentiviral vector was increased less than two-fold. Both free cholesterol and cholesterol ester content in 293-gag-pol producer cells increased with cholesterol addition. In contrast, the phospholipids headgroup composition was essentially unchanged by cholesterol supplementation in 293-gag-pol packaging cells. Based on these results, it is proposed that cholesterol supplementation increases the infectivity of VSV-G-pseudotyped retroviral and lentiviral vectors, possibly by altering the composition of the producer cell membrane where the viral vectors are assembled and bud, and/or by changing the lipid composition of the viral vectors.
Figures


Similar articles
-
Efficiency of onco-retroviral and lentiviral gene transfer into primary mouse and human B-lymphocytes is pseudotype dependent.Hum Gene Ther. 2003 Feb 10;14(3):263-76. doi: 10.1089/10430340360535814. Hum Gene Ther. 2003. PMID: 12639306
-
Introducing a cleavable signal peptide enhances the packaging efficiency of lentiviral vectors pseudotyped with Japanese encephalitis virus envelope proteins.Virus Res. 2017 Feb 2;229:9-16. doi: 10.1016/j.virusres.2016.12.007. Epub 2016 Dec 16. Virus Res. 2017. PMID: 27993624
-
Inducible, high-level production of infectious murine leukemia retroviral vector particles pseudotyped with vesicular stomatitis virus G envelope protein.Hum Gene Ther. 1995 Sep;6(9):1203-13. doi: 10.1089/hum.1995.6.9-1203. Hum Gene Ther. 1995. PMID: 8527479
-
Generation of high-titer pseudotyped retroviral vectors with very broad host range.Methods Cell Biol. 1994;43 Pt A:99-112. doi: 10.1016/s0091-679x(08)60600-7. Methods Cell Biol. 1994. PMID: 7823872 Review.
-
Altering the tropism of lentiviral vectors through pseudotyping.Curr Gene Ther. 2005 Aug;5(4):387-98. doi: 10.2174/1566523054546224. Curr Gene Ther. 2005. PMID: 16101513 Free PMC article. Review.
Cited by
-
Establishment of mouse leukemia cell lines expressing human CD4/CCR5 using lentiviral vectors.Virus Genes. 2017 Apr;53(2):197-204. doi: 10.1007/s11262-016-1423-x. Epub 2016 Dec 28. Virus Genes. 2017. PMID: 28028680
-
Construction and applications of SARS-CoV-2 pseudoviruses: a mini review.Int J Biol Sci. 2021 Apr 10;17(6):1574-1580. doi: 10.7150/ijbs.59184. eCollection 2021. Int J Biol Sci. 2021. PMID: 33907521 Free PMC article. Review.
-
Current status on the development of pseudoviruses for enveloped viruses.Rev Med Virol. 2018 Jan;28(1):e1963. doi: 10.1002/rmv.1963. Epub 2017 Dec 7. Rev Med Virol. 2018. PMID: 29218769 Free PMC article. Review.
-
Roles of Cholesterol in Early and Late Steps of the Nipah Virus Membrane Fusion Cascade.J Virol. 2021 Feb 24;95(6):e02323-20. doi: 10.1128/JVI.02323-20. Print 2021 Feb 24. J Virol. 2021. PMID: 33408170 Free PMC article.
-
Assessment of selected media supplements to improve F/HN lentiviral vector production yields.Sci Rep. 2017 Aug 31;7(1):10198. doi: 10.1038/s41598-017-07893-3. Sci Rep. 2017. PMID: 28860488 Free PMC article.
References
-
- Campbell S.M., Crowe S.M., Mak J. Lipid rafts and HIV-1: from viral entry to assembly of progeny virions. J. Clin. Virol. 2001;22:217–227. - PubMed
-
- Isa P., Realpe M., Romero P., Lopez S., Arias C.F. Rotavirus RRV associates with lipid membrane microdomains during cell entry. Virology. 2004;322:370–381. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
