Charge State Dependent Fragmentation of Gaseous α-Synuclein Cations via Ion Trap and Beam-Type Collisional Activation
- PMID: 20160958
- PMCID: PMC2759116
- DOI: 10.1016/j.ijms.2008.12.007
Charge State Dependent Fragmentation of Gaseous α-Synuclein Cations via Ion Trap and Beam-Type Collisional Activation
Abstract
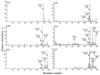
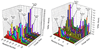
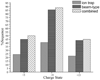
Ions derived from nano-electrospray ionization (nano-ESI) of α-synuclein, a 14.5 kDa, 140 amino acid residue protein that is a major component of the Lewy bodies associated with Parkinson's disease, have been subjected to ion trap and beam-type collisional activation. The former samples products from fragmentation at rates generally lower than 100 s(-1) whereas the latter samples products from fragmentation at rates generally greater than 10(3) s(-1). A wide range of protein charge states spanning from as high as [M+17H](17+) to as low as [M+4H](4+) have been formed either directly from nano-ESI or via ion/ion proton transfer reactions involving the initially formed protein cations and have been subjected to both forms of collision-induced dissociation (CID). The extent of sequence information (i.e., number of distinct amide bond cleavages) available from either CID method was found to be highly sensitive to protein precursor ion charge state. Furthermore, the relative contributions of the various competing dissociation channels were also dependent upon precursor ion charge state. The qualitative trends in the changes in extent of amide bond cleavages and identities of bonds cleaved with precursor ion charge state were similar for two forms of CID. However, for every charge state examined, roughly twice the primary sequence information resulted from beam-type CID relative to ion trap CID. For example, evidence for cleavage of 86% of the protein amide bonds was observed for the [M+9H](9+) precursor ion using beam-type CID whereas 41% of the bonds were cleaved for the same precursor ion using ion trap CID. The higher energies required to drive fragmentation reactions at rates necessary to observe products in the beam experiment access more of the structurally informative fragmentation channels, which has important implications for whole protein tandem mass spectrometry.
Figures





Similar articles
-
Ion trap versus low-energy beam-type collision-induced dissociation of protonated ubiquitin ions.Anal Chem. 2006 Feb 15;78(4):1218-27. doi: 10.1021/ac051622b. Anal Chem. 2006. PMID: 16478115
-
Ion trap collision-induced dissociation of human hemoglobin alpha-chain cations.J Am Soc Mass Spectrom. 2006 Jul;17(7):923-31. doi: 10.1016/j.jasms.2006.01.004. Epub 2006 May 15. J Am Soc Mass Spectrom. 2006. PMID: 16698278
-
On performing simultaneous electron transfer dissociation and collision-induced dissociation on multiply protonated peptides in a linear ion trap.J Am Soc Mass Spectrom. 2009 Sep;20(9):1672-83. doi: 10.1016/j.jasms.2009.05.009. Epub 2009 May 20. J Am Soc Mass Spectrom. 2009. PMID: 19539496
-
Ion/ion chemistry of high-mass multiply charged ions.Mass Spectrom Rev. 1998 Nov-Dec;17(6):369-407. doi: 10.1002/(SICI)1098-2787(1998)17:6<369::AID-MAS1>3.0.CO;2-J. Mass Spectrom Rev. 1998. PMID: 10360331 Review.
-
Ion activation methods for tandem mass spectrometry.J Mass Spectrom. 2004 Oct;39(10):1091-112. doi: 10.1002/jms.703. J Mass Spectrom. 2004. PMID: 15481084 Review.
Cited by
-
Increasing Fragmentation of Disulfide-Bonded Proteins for Top-Down Mass Spectrometry by Supercharging.Int J Mass Spectrom. 2015 Feb 1;377:546-556. doi: 10.1016/j.ijms.2014.07.047. Int J Mass Spectrom. 2015. PMID: 26028988 Free PMC article.
-
Top-down protein identification of proteasome proteins with nanoLC-FT-ICR-MS employing data-independent fragmentation methods.Proteomics. 2014 May;14(10):1271-82. doi: 10.1002/pmic.201300339. Epub 2014 Mar 26. Proteomics. 2014. PMID: 24478249 Free PMC article.
-
Novel Peptide Ion Chemistry Associated with Gold (I) Cationization: Preferential Cleavage at Lysine Residues.Int J Mass Spectrom. 2018 Apr;427:114-122. doi: 10.1016/j.ijms.2017.11.011. Epub 2017 Nov 21. Int J Mass Spectrom. 2018. PMID: 29881326 Free PMC article.
-
Gas-phase chemistry of multiply charged bioions in analytical mass spectrometry.Annu Rev Anal Chem (Palo Alto Calif). 2010;3:365-85. doi: 10.1146/annurev.anchem.111808.073725. Annu Rev Anal Chem (Palo Alto Calif). 2010. PMID: 20636047 Free PMC article. Review.
-
Analysis of intact protein isoforms by mass spectrometry.J Biol Chem. 2011 Jul 22;286(29):25451-8. doi: 10.1074/jbc.R111.239442. Epub 2011 Jun 1. J Biol Chem. 2011. PMID: 21632550 Free PMC article. Review.
References
Grants and funding
LinkOut - more resources
Full Text Sources
