Through-Space Effects of Substituents Dominate Molecular Electrostatic Potentials of Substituted Arenes
- PMID: 20161573
- PMCID: PMC2806064
- DOI: 10.1021/ct900344g
Through-Space Effects of Substituents Dominate Molecular Electrostatic Potentials of Substituted Arenes
Abstract
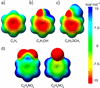
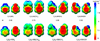
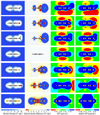
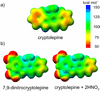
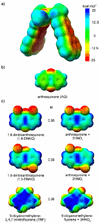
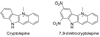
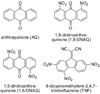
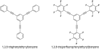
Model systems have been studied using density functional theory to assess the contributions of π-resonance and through-space effects on electrostatic potentials of substituted arenes. The results contradict the widespread assumption that changes in molecular ESPs reflect only local changes in the electron density. Substituent effects on the ESP above the molecular plane are commonly attributed to changes in the aryl π-system. We show that ESP changes for a collection of substituted benzenes and more complex aromatic systems can be accounted for mostly by through-space effects, with no change in the aryl π-electron density. Only when π-resonance effects are substantial do they influence changes in the ESP above the aromatic ring to any extent. Examples of substituted arenes studied here are taken from the fields of drug design, host-guest chemistry, and crystal engineering. These findings emphasize the potential pitfalls of assuming ESP changes reflect changes in the local electron density. Since ESP changes are frequently used to rationalize and predict intermolecular interactions, these findings have profound implications for our understanding of substituent effects in countless areas of chemistry and molecular biology. Specifically, in many non-covalent interactions there are significant, often neglected, through-space interactions with the substituents. Finally, the present results explain the perhaps unexpectedly good performance of many molecular mechanics force-fields applied to supramolecular assembly phenomena and π-π interactions in biological systems despite the neglect of the polarization of the aryl π-system by substituents.
Figures



Similar articles
-
Understanding substituent effects in noncovalent interactions involving aromatic rings.Acc Chem Res. 2013 Apr 16;46(4):1029-38. doi: 10.1021/ar300109n. Epub 2012 Jun 22. Acc Chem Res. 2013. PMID: 22725832
-
Substituent effects in cation/pi interactions and electrostatic potentials above the centers of substituted benzenes are due primarily to through-space effects of the substituents.J Am Chem Soc. 2009 Mar 11;131(9):3126-7. doi: 10.1021/ja809097r. J Am Chem Soc. 2009. PMID: 19219986 Free PMC article.
-
Are anion/pi interactions actually a case of simple charge-dipole interactions?J Phys Chem A. 2010 Aug 26;114(33):8658-64. doi: 10.1021/jp1010549. J Phys Chem A. 2010. PMID: 20433187 Free PMC article.
-
A Supramolecular Approach to Structure-Based Design with A Focus on Synthons Hierarchy in Ornithine-Derived Ligands: Review, Synthesis, Experimental and in Silico Studies.Molecules. 2020 Mar 3;25(5):1135. doi: 10.3390/molecules25051135. Molecules. 2020. PMID: 32138329 Free PMC article. Review.
-
Principles of Cation-π Interactions for Engineering Mussel-Inspired Functional Materials.Acc Chem Res. 2022 Apr 19;55(8):1171-1182. doi: 10.1021/acs.accounts.2c00068. Epub 2022 Mar 28. Acc Chem Res. 2022. PMID: 35344662 Review.
Cited by
-
The interplay between hydrogen and halogen bonding: substituent effects and their role in the hydrogen bond enhanced halogen bond.Chem Sci. 2023 Jul 21;14(33):8924-8935. doi: 10.1039/d3sc02348f. eCollection 2023 Aug 23. Chem Sci. 2023. PMID: 37621436 Free PMC article.
-
Dissection of the Polar and Non-Polar Contributions to Aromatic Stacking Interactions in Solution.Angew Chem Int Ed Engl. 2021 Oct 25;60(44):23871-23877. doi: 10.1002/anie.202110809. Epub 2021 Oct 1. Angew Chem Int Ed Engl. 2021. PMID: 34472177 Free PMC article.
-
Quantifying Through-Space Substituent Effects.Angew Chem Int Ed Engl. 2020 Sep 14;59(38):16717-16724. doi: 10.1002/anie.202006943. Epub 2020 Jul 17. Angew Chem Int Ed Engl. 2020. PMID: 32542910 Free PMC article.
-
Nanoscale Self-Assembly for Therapeutic Delivery.Front Bioeng Biotechnol. 2020 Feb 25;8:127. doi: 10.3389/fbioe.2020.00127. eCollection 2020. Front Bioeng Biotechnol. 2020. PMID: 32158749 Free PMC article. Review.
-
Tunable CH/π Interactions within a Tryptophan Zipper Motif to Stabilize the Fold of Long β-Hairpin Peptides.ACS Chem Biol. 2023 Dec 15;18(12):2555-2563. doi: 10.1021/acschembio.3c00553. Epub 2023 Nov 17. ACS Chem Biol. 2023. PMID: 37976523 Free PMC article.
References
-
- Politzer P, Truhlar DG. Chemical Applications of Atomic and Molecular Electrostatic Potentials. New York: Plenum; 1981.
- Murray JS, Sen K. Molecular Electrostatic Potentials: Concepts and Applications. Elsevier Science; 1996.
- Naráy-Szabó G, Ferenczy GG. Chem. Rev. 1995;95:829–847.
-
- Politzer P, Murray JS. In: Computational Medical Chemistry for Drug Discovery. Bultinck P, De Winter H, Langenaeker W, Tollenaere JP, editors. New York: Marcel Dekker Inc; 2004. pp. 213–234.
-
- Politzer P, Murray JS. In: Reviews in Computational Chemistry. Lipkowitz KB, Boyd DB, editors. Vol. 2. New York: VCH Publishers; 1991.
-
- Meyer EA, Castellano RK, Diederich F. Angew. Chem. Int. Ed. 2003;42:1210–1250. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous
