Sources of Urinary Proteins and their Analysis by Urinary Proteomics for the Detection of Biomarkers of Disease
- PMID: 20161589
- PMCID: PMC2808139
- DOI: 10.1002/prca.200800243
Sources of Urinary Proteins and their Analysis by Urinary Proteomics for the Detection of Biomarkers of Disease
Abstract
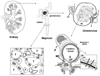
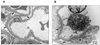
Renal disorders account for a substantial fraction of the budget for health care in many countries. Proteinuria is a frequent manifestation in afflicted patients, but the origin of the proteins varies based on the nature of the disorder. The emerging field of urinary proteomics has the potential to replace kidney biopsy as the diagnostic procedure of choice for patients with some glomerular forms of renal disease. To fully realize this potential, it is vital to understand the basis for the urinary excretion of protein in physiological and pathological conditions. In this review, we discuss the structure of the nephron, the functional unit of the kidney, and the process by which proteins/peptides enter the urine. We discuss several aspects of proteinuria that impact the proteomic analysis of urine of patients with renal diseases.
Conflict of interest statement
The authors do not have any financial/commercial conflicts of interest to declare.
Figures
References
-
- Briggs JP, Kriz W, Schnermann JB. In: Primer on Kidney Disease. Greenberg A, editor. Philadelphia: Elsevier Saunders; 2005. pp. 2–19.
-
- Rostgaard J, Qvortrup K. Sieve plugs in fenestrae of glomerular capillaries--site of the filtration barrier? Cells Tissues Organs. 2002;170:132–138. - PubMed
-
- Deen WM, Lazzara MJ, Myers BD. Structural determinants of glomerular permeability. Am. J. Physiol. Renal Physiol. 2001;281:F579–F596. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

