Synthesis of Substituted Quinolines by the Electrophilic Cyclization of N-(2-Alkynyl)anilines
- PMID: 20161633
- PMCID: PMC2813516
- DOI: 10.1016/j.tet.2009.12.012
Synthesis of Substituted Quinolines by the Electrophilic Cyclization of N-(2-Alkynyl)anilines
Abstract
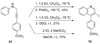
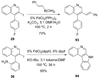
A wide variety of substituted quinolines are readily synthesized under mild reaction conditions by the 6-endo-dig electrophilic cyclization of N-(2-alkynyl)anilines by ICl, I(2), Br(2), PhSeBr and p-O(2)NC(6)H(4)SCl. The reaction affords 3-halogen-, selenium- and sulfur-containing quinolines in moderate to good yields in the presence of various functional groups. Analogous quinolines bearing a hydrogen in the 3-position have been synthesized by the Hg(OTf)(2)-catalyzed ring closure of these same alkynylanilines.
Figures
Similar articles
-
Synthesis of substituted isoquinolines by electrophilic cyclization of iminoalkynes.J Org Chem. 2002 May 17;67(10):3437-44. doi: 10.1021/jo020020e. J Org Chem. 2002. PMID: 12003556
-
Synthesis of 5-iodopyrrolo[1,2-a]quinolines and indolo[1,2-a]quinolines via iodine-mediated electrophilic and regioselective 6-endo-dig ring closure.J Org Chem. 2011 Jul 15;76(14):5670-84. doi: 10.1021/jo200638k. Epub 2011 Jun 16. J Org Chem. 2011. PMID: 21619026
-
Synthesis of substituted quinolines by electrophilic cyclization of N-(2-alkynyl)anilines.Org Lett. 2005 Mar 3;7(5):763-6. doi: 10.1021/ol0476218. Org Lett. 2005. PMID: 15727435
-
Synthesis of naphthalenes and 2-naphthols by the electrophilic cyclization of alkynes.J Org Chem. 2006 Jan 6;71(1):236-43. doi: 10.1021/jo051948k. J Org Chem. 2006. PMID: 16388642 Free PMC article.
-
5-Endo-dig cyclizations in organic syntheses.Mol Divers. 2020 Feb;24(1):295-317. doi: 10.1007/s11030-019-09930-x. Epub 2019 Mar 5. Mol Divers. 2020. PMID: 30838496 Review.
Cited by
-
Synthesis and crystal structures of (E)-N'-(4-chloro-3-nitro-benzyl-idene)acetohydrazide and (E)-2-(4-chloro-benzyl-idene)-1-(quinolin-8-yl)hydrazine.Acta Crystallogr E Crystallogr Commun. 2023 Jul 28;79(Pt 8):762-766. doi: 10.1107/S2056989023006412. eCollection 2023 Jul 1. Acta Crystallogr E Crystallogr Commun. 2023. PMID: 37601392 Free PMC article.
-
Palladium-catalyzed 1,4-difunctionalization of butadiene to form skipped polyenes.J Am Chem Soc. 2013 Mar 20;135(11):4167-70. doi: 10.1021/ja3110544. Epub 2013 Mar 12. J Am Chem Soc. 2013. PMID: 23473452 Free PMC article.
-
Rational Design and Facile Synthesis of a Highly Tunable Quinoline-Based Fluorescent Small-Molecule Scaffold for Live Cell Imaging.J Am Chem Soc. 2018 Aug 1;140(30):9486-9493. doi: 10.1021/jacs.8b03738. Epub 2018 Jul 20. J Am Chem Soc. 2018. PMID: 30028130 Free PMC article.
-
Copper-Catalyzed Heteroarylboration of 1,3-Dienes with 3-Bromopyridines: A cine Substitution.Angew Chem Int Ed Engl. 2018 May 22;57(21):6146-6149. doi: 10.1002/anie.201801139. Epub 2018 Apr 26. Angew Chem Int Ed Engl. 2018. PMID: 29697174 Free PMC article.
-
Recent Advances in Metal-Free Quinoline Synthesis.Molecules. 2016 Jul 29;21(8):986. doi: 10.3390/molecules21080986. Molecules. 2016. PMID: 27483222 Free PMC article. Review.
References
-
- Michael JP. Nat. Prod. Rep. 1997;14:605. - PubMed
- Balasubramanian M, Keay JG. In: Comprehensive Heterocyclic Chemistry II. Katritzky AR, Rees CW, Scriven EFV, editors. Vol. 5. Oxford: Pergamon Press; 1996. pp. 245–266.
-
- Newhouse BJ, Bordner J, Augeri DJ, Litts CS, Kleinman EF. J. Org. Chem. 1992;57:6991.
- Torii S, Xu LH, Sadakane M, Okumoto H. Synlett. 1992:513.
- Nobuhide M, Yoshinobu Y, Hiroshi I, Yoshio O, Tamejiro H. Tetrahedron Lett. 1993;24:8263.
- Croisey-Delcey M, Croisy A, Carrez D, Huel C, Chiaroni A, Ducrot P, Bisagni E, Jin L, Leclercq G. Bioorg. Med. Chem. 2000;8:2629. - PubMed
- Bu XY, Deady LW, Denny WA. Aust. J. Chem. 2000;53:143.
- Blackburn TP, Cox B, Guildord AJ, LeCount DJ, Middlemiss DN, Pearce RJ, Thornber CW. J. Med. Chem. 1987;30:2252. - PubMed
- Amii H, Kishikawa Y, Uneyama K. Org. Lett. 2001;3:1109. - PubMed
-
- O’Dell DK, Nicholas KM. J. Org. Chem. 2003;68:6427. and references therein. - PubMed
-
- Eisch JJ. J. Org. Chem. 1961;27:1318.
- Kress TJ, Constantino SM. J. Heterocycl. Chem. 1973;10:409.
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous