B-cell tolerance in transplantation: is repertoire remodeling the answer?
- PMID: 20161663
- PMCID: PMC2819040
- DOI: 10.1586/eci.09.63
B-cell tolerance in transplantation: is repertoire remodeling the answer?
Abstract
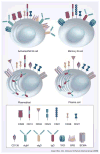
T lymphocytes are the primary targets of immunotherapy in clinical transplantation; however, B lymphocytes and their secreted alloantibodies are also highly detrimental to the allograft. Therefore, the achievement of sustained organ transplant survival will likely require the induction of B-lymphocyte tolerance. During development, acquisition of B-cell tolerance to self-antigens relies on clonal deletion in the early stages of B-cell compartment ontogeny. We contend that this mechanism should be recapitulated in the setting of alloantigens and organ transplantation to eliminate the alloreactive B-cell subset from the recipient. Clinically feasible targets of B-cell-directed immunotherapy, such as CD20 and B-lymphocyte stimulator (BLyS), should drive upcoming clinical trials aimed at remodeling the recipient B-cell repertoire.
Figures


Similar articles
-
B-lymphocyte homeostasis and BLyS-directed immunotherapy in transplantation.Transplant Rev (Orlando). 2010 Oct;24(4):207-21. doi: 10.1016/j.trre.2010.05.004. Epub 2010 Jul 23. Transplant Rev (Orlando). 2010. PMID: 20655723 Free PMC article. Review.
-
Essential role for B cells in transplantation tolerance.Curr Opin Immunol. 2011 Oct;23(5):685-91. doi: 10.1016/j.coi.2011.07.011. Curr Opin Immunol. 2011. PMID: 21982511 Free PMC article. Review.
-
Acquisition of humoral transplantation tolerance upon de novo emergence of B lymphocytes.J Immunol. 2011 Jan 1;186(1):614-20. doi: 10.4049/jimmunol.1002873. Epub 2010 Nov 17. J Immunol. 2011. PMID: 21084661
-
Murine islet allograft tolerance upon blockade of the B-lymphocyte stimulator, BLyS/BAFF.Transplantation. 2012 Apr 15;93(7):676-85. doi: 10.1097/TP.0b013e318246621d. Transplantation. 2012. PMID: 22262127
-
Primary B cell repertoire remodeling to achieve humoral transplantation tolerance.Semin Immunol. 2012 Apr;24(2):109-14. doi: 10.1016/j.smim.2011.08.016. Epub 2011 Oct 5. Semin Immunol. 2012. PMID: 21978627 Review.
Cited by
-
The role of B cells in transplantation and immunopathic diseases.Immune Netw. 2010 Jun;10(3):81-4. doi: 10.4110/in.2010.10.3.81. Epub 2010 Jun 30. Immune Netw. 2010. PMID: 20631877 Free PMC article.
-
Resolve, revise, and relax: the 3 Rs of B cell repertoire adjustment.Immunol Lett. 2012 Mar 30;143(1):2-8. doi: 10.1016/j.imlet.2012.01.014. Epub 2012 Feb 6. Immunol Lett. 2012. PMID: 22330846 Free PMC article. Review.
-
B-lymphocyte homeostasis and BLyS-directed immunotherapy in transplantation.Transplant Rev (Orlando). 2010 Oct;24(4):207-21. doi: 10.1016/j.trre.2010.05.004. Epub 2010 Jul 23. Transplant Rev (Orlando). 2010. PMID: 20655723 Free PMC article. Review.
-
The Positive Impact of Donor Bone Marrow Cells Transplantation into Immunoprivileged Compartments on the Survival of Vascularized Skin Allografts.Arch Immunol Ther Exp (Warsz). 2021 Oct 11;69(1):28. doi: 10.1007/s00005-021-00631-8. Arch Immunol Ther Exp (Warsz). 2021. PMID: 34633538 Free PMC article.
-
Essential role for B cells in transplantation tolerance.Curr Opin Immunol. 2011 Oct;23(5):685-91. doi: 10.1016/j.coi.2011.07.011. Curr Opin Immunol. 2011. PMID: 21982511 Free PMC article. Review.
References
-
- Brennan DC, Daller JA, Lake KD, Cibrik D, Del Castillo D. Rabbit antithymocyte globulin versus basiliximab in renal transplantation. N Engl J Med. 2006;355(19):1967–1977. - PubMed
-
- Pirsch JD, Miller J, Deierhoi MH, Vincenti F, Filo RS. A comparison of tacrolimus (FK506) and cyclosporine for immunosuppression after cadaveric renal transplantation. FK506 Kidney Transplant Study Group. Transplantation. 1997;63(7):977–983. - PubMed
-
- Deeks ED, Keating GM. Rabbit antithymocyte globulin (thymoglobulin): a review of its use in the prevention and treatment of acute renal allograft rejection. Drugs. 2009;69(11):1483–1512. - PubMed
-
- Scherer MN, Banas B, Mantouvalou K, et al. Current concepts and perspectives of immunosuppression in organ transplantation. Langenbecks Arch Surg. 2007;392(5):511–523. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
