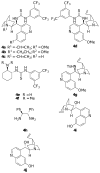
Organocatalyzed synthesis of 2-amino-8-oxo-5,6,7,8-tetrahydro-4H-chromene-3-carbonitriles
- PMID: 20161684
- PMCID: PMC2821098
- DOI: 10.1016/j.tetlet.2009.12.139
Organocatalyzed synthesis of 2-amino-8-oxo-5,6,7,8-tetrahydro-4H-chromene-3-carbonitriles
Abstract
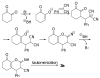
2-Amino-8-oxo-tetrahydro-4H-chromene-3-carbonitriles were synthesized for the first time from a tandem Michael addition - cyclization reaction between cyclohexane-1,2-dione and benzylidenemalononitriles. An enantioselective synthesis of these compounds was achieved in moderate ee values (up to 63% ee) by using a cinchona alkaloid-derived thiouea catalyst.
Figures
Similar articles
-
Enantioselective synthesis of spiro[4H-pyran-3,3'-oxindole] derivatives catalyzed by cinchona alkaloid thioureas: Significant water effects on the enantioselectivity.Synth Commun. 2019;49(21):2971-2982. doi: 10.1080/00397911.2019.1651866. Epub 2019 Aug 14. Synth Commun. 2019. PMID: 33012850 Free PMC article.
-
Organocatalyzed enantioselective synthesis of 2-amino-5-oxo-5,6,7,8-tetrahydro-4H-chromene-3-carboxylates.Tetrahedron Lett. 2011 Dec 14;52(50):6792-6795. doi: 10.1016/j.tetlet.2011.10.040. Tetrahedron Lett. 2011. PMID: 22081731 Free PMC article.
-
Using Sodium Hydride and Potassium Carbonate as Bases in Synthesis of Substituted 2-Amino-4-aryl-7-propargyloxy-4H-chromene-3-carbonitriles.Curr Org Synth. 2019;16(3):423-430. doi: 10.2174/1570179416666190104124652. Curr Org Synth. 2019. PMID: 31984904
-
Organocatalyzed enantioselective synthesis of 6-amino-5-cyanodihydropyrano[2,3-c]pyrazoles.Tetrahedron Lett. 2009 May 13;50(19):2252-2255. doi: 10.1016/j.tetlet.2009.02.210. Tetrahedron Lett. 2009. PMID: 19915654 Free PMC article.
-
Asymmetric synthesis of spiro[4H-chromene-3,3'-oxindoles] via a squaramide-organocatalytic three-component cascade Knoevenagel/Michael/cyclization sequence.Mol Divers. 2025 Feb;29(1):293-302. doi: 10.1007/s11030-024-10852-6. Epub 2024 Apr 30. Mol Divers. 2025. PMID: 38687399
Cited by
-
2-(4-Nitro-benzyl-idene)malononitrile.Acta Crystallogr Sect E Struct Rep Online. 2012 Apr 1;68(Pt 4):o957. doi: 10.1107/S1600536812008896. Epub 2012 Mar 3. Acta Crystallogr Sect E Struct Rep Online. 2012. PMID: 22590017 Free PMC article.
-
K2CO3-mediated synthesis of functionalised 4-substituted-2-amino-3-cyano-4H-chromenes via Michael-cyclization reactions.Molecules. 2014 Nov 25;19(12):19253-68. doi: 10.3390/molecules191219253. Molecules. 2014. PMID: 25429557 Free PMC article.
-
Asymmetric organocatalytic Michael addition of cyclopentane-1,2-dione to alkylidene oxindole.Beilstein J Org Chem. 2022 Feb 3;18:167-173. doi: 10.3762/bjoc.18.18. eCollection 2022. Beilstein J Org Chem. 2022. PMID: 35186153 Free PMC article.
-
Enantioselective synthesis of bicylco[3.2.1]octan-8-ones using a tandem Michael-Henry reaction.Tetrahedron. 2010 Jun 19;66(25):4423-4427. doi: 10.1016/j.tet.2010.04.044. Tetrahedron. 2010. PMID: 20532185 Free PMC article.
-
Enantioselective synthesis of spiro[4H-pyran-3,3'-oxindole] derivatives catalyzed by cinchona alkaloid thioureas: Significant water effects on the enantioselectivity.Synth Commun. 2019;49(21):2971-2982. doi: 10.1080/00397911.2019.1651866. Epub 2019 Aug 14. Synth Commun. 2019. PMID: 33012850 Free PMC article.
References
-
- Foloppe N, Fisher LM, Howes R, Potter A, Robertson AGS, Surgenor AE. Bioorg Med Chem. 2006;14:4792–4802. - PubMed
- Wang JL, Liu D, Zhang ZJ, Shan S, Han X, Srinivasula SM, Croce CM, Alnemri ES, Huang Z. Proc Natl Acad Sci USA. 2000;97:7124–7129. - PMC - PubMed
- El-Tamany ES, El-Shahed FA, Mohamed BH. J Serb Chem Soc. 1999;64:9–18.
- Zaki MEA, Soliman HA, Hiekal OA, Rashad AE. Z Naturforsch C. 2006;61:1–5. - PubMed
- Ismail ZH, Aly GM, El-Degwi MS, Heiba HI, Ghorab MM. Egypt J Biotech. 2003;13:73–82.
-
- Konishi K, Kuragano T, Nohara A. Nippon Noyaku Gakkaishi. 1990;15:241–244.
- Liao SY, Qian L, Miao TF, Shen Y, Zheng KC. J Theor Comput Chem. 2009;8:143–155.
- Kemnitzer W, Jiang S, Wang Y, Kasibhatla S, Crogan-Grundy C, Bubenik M, Labrecque D, Denis R, Lamothe S, Attardo G, Gourdeau H, Tseng B, Drewe J, Cai SX. Bioorg Med Chem Lett. 2008;18:603–607. - PubMed
- Kemnitzer W, Drewe J, Jiang S, Zhang H, Crogan-Grundy C, Labreque D, Bubenick M, Attardo G, Denis R, Lamothe S, Gourdeau H, Tseng B, Kasibhatla S, Cai SX. J Med Chem. 2008;51:417–423. - PubMed
- Bonsignore L, Loy G, Secci D, Calignano A. Eur J Med Chem. 1993;28:517–520.
- Andreani LL, Lapi E. Boll Chim Farm. 1960;99:583–586. - PubMed
-
- Hafez EAA, Elnagdi MH, Elagamey AGA, Ei-Taweel FMAA. Heterocycles. 1987;26:903–907.
- Witte EC, Neubert P, Roesch A. Ger Offen DE3427985. 1986.
- Chem Abstr. 1986;104:224915.
- Morinaka Y, Takahashi K. Jpn Kokai Tokkyo Koho JP52017498. 1977.
- Chem Abstr. 1977;87:102299.
-
- Bloxham J, Dell CP, Smith CW. Heterocycles. 1994;38:399–408.
- Elagamey AGA, Sawllim SZ, El-Taweel FMA, Elnagdi MH. Collect Czech Chem Commun. 1988;53:1534–1538.
- Ballini R, Bosica G, Conforti ML, Maggi R, Mazzacani A, Righi P, Sartori G. Tetrahedron. 2001;57:1395–1398.
- Jin TS, Zhang JS, Liu LB, Wang AQ, Li TS. Synth Commun. 2006;36:2009–2015.
- Zhang AQ, Zhang M, Chen HH, Chen J, Chen HY. Synth Commun. 2007;37:231–235.
Grants and funding
LinkOut - more resources
Full Text Sources