New class of monoclonal antibodies against severe influenza: prophylactic and therapeutic efficacy in ferrets
- PMID: 20161706
- PMCID: PMC2817000
- DOI: 10.1371/journal.pone.0009106
New class of monoclonal antibodies against severe influenza: prophylactic and therapeutic efficacy in ferrets
Abstract
Background: The urgent medical need for innovative approaches to control influenza is emphasized by the widespread resistance of circulating subtype H1N1 viruses to the leading antiviral drug oseltamivir, the pandemic threat posed by the occurrences of human infections with highly pathogenic avian H5N1 viruses, and indeed the evolving swine-origin H1N1 influenza pandemic. A recently discovered class of human monoclonal antibodies with the ability to neutralize a broad spectrum of influenza viruses (including H1, H2, H5, H6 and H9 subtypes) has the potential to prevent and treat influenza in humans. Here we report the latest efficacy data for a representative antibody of this novel class.
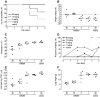
Methodology/principal findings: We evaluated the prophylactic and therapeutic efficacy of the human monoclonal antibody CR6261 against lethal challenge with the highly pathogenic avian H5N1 virus in ferrets, the optimal model of human influenza infection. Survival rates, clinically relevant disease signs such as changes in body weight and temperature, virus replication in lungs and upper respiratory tract, as well as macro- and microscopic pathology were investigated. Prophylactic administration of 30 and 10 mg/kg CR6261 prior to viral challenge completely prevented mortality, weight loss and reduced the amount of infectious virus in the lungs by more than 99.9%, abolished shedding of virus in pharyngeal secretions and largely prevented H5N1-induced lung pathology. When administered therapeutically 1 day after challenge, 30 mg/kg CR6261 prevented death in all animals and blunted disease, as evidenced by decreased weight loss and temperature rise, reduced lung viral loads and shedding, and less lung damage.
Conclusions/significance: These data demonstrate the prophylactic and therapeutic efficacy of this new class of human monoclonal antibodies in a highly stringent and clinically relevant animal model of influenza and justify clinical development of this approach as intervention for both seasonal and pandemic influenza.
Conflict of interest statement
Figures

References
-
- WHO. Fact sheet 211: Influenza. World Health Organization. 2009.
-
- Hoelscher M, Gangappa S, Zhong W, Jayashankar L, Sambhara S. Vaccines against epidemic and pandemic influenza. Expert Opin Drug Deliv. 2008;5:1139–1157. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

