DKK1 mediated inhibition of Wnt signaling in postnatal mice leads to loss of TEC progenitors and thymic degeneration
- PMID: 20161711
- PMCID: PMC2817005
- DOI: 10.1371/journal.pone.0009062
DKK1 mediated inhibition of Wnt signaling in postnatal mice leads to loss of TEC progenitors and thymic degeneration
Abstract
Background: Thymic epithelial cell (TEC) microenvironments are essential for the recruitment of T cell precursors from the bone marrow, as well as the subsequent expansion and selection of thymocytes resulting in a mature self-tolerant T cell repertoire. The molecular mechanisms, which control both the initial development and subsequent maintenance of these critical microenvironments, are poorly defined. Wnt signaling has been shown to be important to the development of several epithelial tissues and organs. Regulation of Wnt signaling has also been shown to impact both early thymocyte and thymic epithelial development. However, early blocks in thymic organogenesis or death of the mice have prevented analysis of a role of canonical Wnt signaling in the maintenance of TECs in the postnatal thymus.
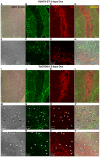
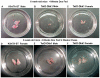
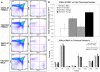
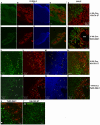
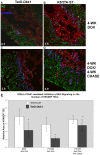
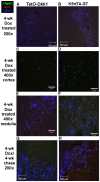
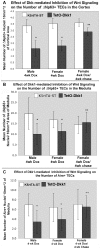
Methodology/principal findings: Here we demonstrate that tetracycline-regulated expression of the canonical Wnt inhibitor DKK1 in TECs localized in both the cortex and medulla of adult mice, results in rapid thymic degeneration characterized by a loss of DeltaNP63(+) Foxn1(+) and Aire(+) TECs, loss of K5K8DP TECs thought to represent or contain an immature TEC progenitor, decreased TEC proliferation and the development of cystic structures, similar to an aged thymus. Removal of DKK1 from DKK1-involuted mice results in full recovery, suggesting that canonical Wnt signaling is required for the differentiation or proliferation of TEC populations needed for maintenance of properly organized adult thymic epithelial microenvironments.
Conclusions/significance: Taken together, the results of this study demonstrate that canonical Wnt signaling within TECs is required for the maintenance of epithelial microenvironments in the postnatal thymus, possibly through effects on TEC progenitor/stem cell populations. Downstream targets of Wnt signaling, which are responsible for maintenance of these TEC progenitors may provide useful targets for therapies aimed at counteracting age associated thymic involution or the premature thymic degeneration associated with cancer therapy and bone marrow transplants.
Conflict of interest statement
Figures


Similar articles
-
Abrogation of Notch Signaling in Embryonic TECs Impacts Postnatal mTEC Homeostasis and Thymic Involution.Front Immunol. 2022 May 30;13:867302. doi: 10.3389/fimmu.2022.867302. eCollection 2022. Front Immunol. 2022. PMID: 35707539 Free PMC article.
-
Regeneration of the adult thymus is preceded by the expansion of K5+K8+ epithelial cell progenitors and by increased expression of Trp63, cMyc and Tcf3 transcription factors in the thymic stroma.Int Immunol. 2007 Nov;19(11):1249-60. doi: 10.1093/intimm/dxm092. Epub 2007 Sep 6. Int Immunol. 2007. PMID: 17823311
-
The Wnt signaling antagonist Kremen1 is required for development of thymic architecture.Clin Dev Immunol. 2006 Jun-Dec;13(2-4):299-319. doi: 10.1080/17402520600935097. Clin Dev Immunol. 2006. PMID: 17162372 Free PMC article.
-
Adult thymic epithelial cell (TEC) progenitors and TEC stem cells: Models and mechanisms for TEC development and maintenance.Eur J Immunol. 2015 Nov;45(11):2985-93. doi: 10.1002/eji.201545844. Epub 2015 Sep 30. Eur J Immunol. 2015. PMID: 26362014 Review.
-
Molecular regulatory networks of thymic epithelial cell differentiation.Differentiation. 2019 May-Jun;107:42-49. doi: 10.1016/j.diff.2019.06.002. Epub 2019 Jun 14. Differentiation. 2019. PMID: 31238242 Review.
Cited by
-
β-catenin/TCF-1 pathway in T cell development and differentiation.J Neuroimmune Pharmacol. 2012 Dec;7(4):750-62. doi: 10.1007/s11481-012-9367-y. Epub 2012 Apr 27. J Neuroimmune Pharmacol. 2012. PMID: 22535304 Free PMC article. Review.
-
Loss of epidermal Evi/Wls results in a phenotype resembling psoriasiform dermatitis.J Exp Med. 2013 Aug 26;210(9):1761-77. doi: 10.1084/jem.20121871. Epub 2013 Aug 5. J Exp Med. 2013. PMID: 23918954 Free PMC article.
-
Label retention identifies a multipotent mesenchymal stem cell-like population in the postnatal thymus.PLoS One. 2013 Dec 10;8(12):e83024. doi: 10.1371/journal.pone.0083024. eCollection 2013. PLoS One. 2013. PMID: 24340075 Free PMC article.
-
Thymus involution and regeneration: two sides of the same coin?Nat Rev Immunol. 2013 Nov;13(11):831-8. doi: 10.1038/nri3534. Epub 2013 Sep 20. Nat Rev Immunol. 2013. PMID: 24052146 Review.
-
Dickkopf proteins in pathological inflammatory diseases.J Leukoc Biol. 2022 Apr;111(4):893-901. doi: 10.1002/JLB.3RI0721-385R. Epub 2021 Dec 10. J Leukoc Biol. 2022. PMID: 34890067 Free PMC article. Review.
References
-
- Anderson G, Jenkinson EJ. Lymphostromal interactions in thymic development and function. Nat Rev Immunol. 2001;1:31–40. - PubMed
-
- Savage PA, Davis MM. A kinetic window constricts the T cell receptor repertoire in the thymus. Immunity. 2001;14:243–252. - PubMed
-
- van Ewijk W, Wang B, Hollander G, Kawamoto H, Spanopoulou E, et al. Thymic microenvironments, 3-D versus 2-D? Semin Immunol. 1999;11:57–64. - PubMed
-
- van Ewijk W, Hollander G, Terhorst C, Wang B. Stepwise development of thymic microenvironments in vivo is regulated by thymocyte subsets. Development. 2000;127:1583–1591. - PubMed
-
- Itoi M, Tsukamoto N, Yoshida H, Amagai T. Mesenchymal cells are required for functional development of thymic epithelial cells. Int Immunol. 2007;19:953–964. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- U54 1U54CA137788-01/CA/NCI NIH HHS/United States
- G12 MD007603/MD/NIMHD NIH HHS/United States
- G12 RR003060/RR/NCRR NIH HHS/United States
- SC1 AI104994/AI/NIAID NIH HHS/United States
- U54 CA132378/CA/NCI NIH HHS/United States
- R01-DE015342/DE/NIDCR NIH HHS/United States
- U56 CA096299/CA/NCI NIH HHS/United States
- 1SC1AI081527/AI/NIAID NIH HHS/United States
- R01-HD053829/HD/NICHD NIH HHS/United States
- R01 DE015342/DE/NIDCR NIH HHS/United States
- R01-AR47709/AR/NIAMS NIH HHS/United States
- U54 CA137788/CA/NCI NIH HHS/United States
- R01 AR047709/AR/NIAMS NIH HHS/United States
- SC1 AI081527/AI/NIAID NIH HHS/United States
- 1U56 CA96299/CA/NCI NIH HHS/United States
- R01 HD053829/HD/NICHD NIH HHS/United States
- G12RR03060/RR/NCRR NIH HHS/United States
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

