Human cord blood-derived AC133+ progenitor cells preserve endothelial progenitor characteristics after long term in vitro expansion
- PMID: 20161785
- PMCID: PMC2820083
- DOI: 10.1371/journal.pone.0009173
Human cord blood-derived AC133+ progenitor cells preserve endothelial progenitor characteristics after long term in vitro expansion
Abstract
Background: Stem cells/progenitors are central to the development of cell therapy approaches for vascular ischemic diseases. The crucial step in rescuing tissues from ischemia is improvement of vascularization that can be achieved by promoting neovascularization. Endothelial progenitor cells (EPCs) are the best candidates for developing such an approach due to their ability to self-renew, circulate and differentiate into mature endothelial cells (ECs). Studies showed that intravenously administered progenitors isolated from bone marrow, peripheral or cord blood home to ischemic sites. However, the successful clinical application of such transplantation therapy is limited by low quantities of EPCs that can be generated from patients. Hence, the ability to amplify the numbers of autologous EPCs by long term in vitro expansion while preserving their angiogenic potential is critically important for developing EPC based therapies. Therefore, the objective of this study was to evaluate the capacity of cord blood (CB)-derived AC133+ cells to differentiate, in vitro, towards functional, mature endothelial cells (ECs) after long term in vitro expansion.
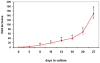
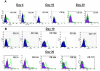
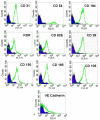
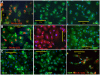
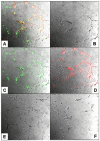
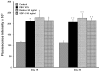
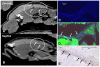
Methodology: We systematically characterized the properties of CB AC133+ cells over the 30 days of in vitro expansion. During 30 days of culturing, CB AC133+ cells exhibited significant growth potential that was manifested as 148-fold increase in cell numbers. Flow cytometry and immunocytochemistry demonstrated that CB AC133+ cells' expression of endothelial progenitor markers was not affected by long term in vitro culturing. After culturing under EC differentiation conditions, cells exhibited high expression of mature ECs markers, such as CD31, VEGFR-2 and von Willebrand factor, as well as the morphological changes indicative of differentiation towards mature ECs. In addition, throughout the 30 day culture cells preserved their functional capacity that was demonstrated by high uptake of DiI fluorescently conjugated-acetylated-low density lipoprotein (DiI-Ac-LDL), in vitro and in vivo migration towards chemotactic stimuli and in vitro tube formation.
Conclusions: These studies demonstrate that primary CB AC133+ culture contained mainly EPCs and that long term in vitro conditions facilitated the maintenance of these cells in the state of commitment towards endothelial lineage.
Conflict of interest statement
Figures

Similar articles
-
MRI tracking of FePro labeled fresh and cryopreserved long term in vitro expanded human cord blood AC133+ endothelial progenitor cells in rat glioma.PLoS One. 2012;7(5):e37577. doi: 10.1371/journal.pone.0037577. Epub 2012 May 25. PLoS One. 2012. PMID: 22662174 Free PMC article.
-
Enhancement of neovascularization with cord blood CD133+ cell-derived endothelial progenitor cell transplantation.Thromb Haemost. 2004 Jun;91(6):1202-12. doi: 10.1160/TH03-06-0378. Thromb Haemost. 2004. PMID: 15175808
-
Immunological and ultrastructural characterization of endothelial cell cultures differentiated from human cord blood derived endothelial progenitor cells.Histochem Cell Biol. 2006 Dec;126(6):649-64. doi: 10.1007/s00418-006-0201-6. Epub 2006 Jun 10. Histochem Cell Biol. 2006. PMID: 16767408
-
Endothelial progenitor cells in angiogenesis.Sheng Li Xue Bao. 2005 Feb 25;57(1):1-6. Sheng Li Xue Bao. 2005. PMID: 15719128 Review.
-
Endothelial progenitor cells: mobilization, differentiation, and homing.Arterioscler Thromb Vasc Biol. 2003 Jul 1;23(7):1185-9. doi: 10.1161/01.ATV.0000073832.49290.B5. Epub 2003 Apr 24. Arterioscler Thromb Vasc Biol. 2003. PMID: 12714439 Review.
Cited by
-
The IMMENSE Study: The Interplay Between iMMune and ENdothelial Cells in Mediating Cardiovascular Risk in Systemic Lupus Erythematosus.Front Immunol. 2020 Oct 29;11:572876. doi: 10.3389/fimmu.2020.572876. eCollection 2020. Front Immunol. 2020. PMID: 33193356 Free PMC article.
-
CD34+ VEGFR-3+ progenitor cells have a potential to differentiate towards lymphatic endothelial cells.J Cell Mol Med. 2014 Mar;18(3):422-33. doi: 10.1111/jcmm.12233. Epub 2014 Jan 22. J Cell Mol Med. 2014. PMID: 24450475 Free PMC article.
-
Differential gene expression in Lin-/VEGF-R2+ bone marrow-derived endothelial progenitor cells isolated from diabetic mice.Cardiovasc Diabetol. 2014 Feb 12;13:42. doi: 10.1186/1475-2840-13-42. Cardiovasc Diabetol. 2014. PMID: 24521356 Free PMC article.
-
Immunomagnetic Delivery of Adipose-Derived Endothelial Progenitor Cells for the Repair of Renal Ischemia-Reperfusion Injury in a Rat Model.Bioengineering (Basel). 2023 Apr 24;10(5):509. doi: 10.3390/bioengineering10050509. Bioengineering (Basel). 2023. PMID: 37237579 Free PMC article.
-
Vascular Mimicry: A Novel Neovascularization Mechanism Driving Anti-Angiogenic Therapy (AAT) Resistance in Glioblastoma.Transl Oncol. 2017 Aug;10(4):650-660. doi: 10.1016/j.tranon.2017.04.007. Epub 2017 Jun 29. Transl Oncol. 2017. PMID: 28668763 Free PMC article. Review.
References
-
- Carmeliet P. Angiogenesis in life, disease and medicine. Nature. 2005;438:932–936. - PubMed
-
- Risau W, Flamme I. Vasculogenesis. Annu Rev Cell Dev Biol. 1995;11:73–91. - PubMed
-
- Risau W, Sariola H, Zerwes HG, Sasse J, Ekblom P, et al. Vasculogenesis and angiogenesis in embryonic-stem-cell-derived embryoid bodies. Development. 1988;102:471–478. - PubMed
-
- Folkman J. Seminars in Medicine of the Beth Israel Hospital, Boston. Clinical applications of research on angiogenesis. N Engl J Med. 1995;333:1757–1763. - PubMed
-
- Folkman J, Shing Y. Angiogenesis. J Biol Chem. 1992;267:10931–10934. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

