On the stereochemical course of palladium-catalyzed cross-coupling of allylic silanolate salts with aromatic bromides
- PMID: 20163185
- PMCID: PMC2836786
- DOI: 10.1021/ja910804u
On the stereochemical course of palladium-catalyzed cross-coupling of allylic silanolate salts with aromatic bromides
Abstract
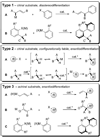
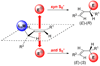
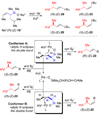
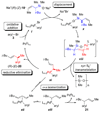
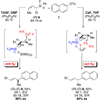
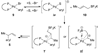
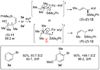
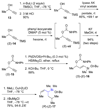
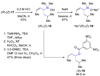
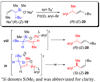
The stereochemical course of palladium-catalyzed cross-coupling reactions of an enantioenriched, alpha-substituted, allylic silanolate salt with aromatic bromides has been investigated. The allylic silanolate salt was prepared in high geometrical (Z/E, 94:6) and high enantiomeric (94:6 er) purity by a copper-catalyzed S(N)2' reaction of a resolved allylic carbamate. Eight different aromatic bromides underwent cross-coupling with excellent constitutional site-selectivity and excellent stereospecificity. Stereochemical correlation established that the transmetalation event proceeds through a syn S(E)' mechanism which is interpreted in terms of an intramolecular delivery of the arylpalladium electrophile through a key intermediate that contains a discrete Si-O-Pd linkage.
Figures


References
-
- de Meijere A, Diederich F, editors. Metal-Catalyzed Cross-Coupling Reactions. Second Ed. Weinheim: Wiley-VCH; 2004.
- Negishi E-I, editor. Handbook of Organopalladium Chemistry for Organic Synthesis. New York: Wiley–Interscience; 2002.
-
- Ojima I, editor. Catalytic Asymmetric Synthesis. Second Ed. New York: Wiley–VCH; 2000.
- Tietze LF, Ila H, Bell HP. Chem. Rev. 2004;104:3453–3516. - PubMed
- Hayashi TJ. Organomet. Chem. 2002;653:41–45.
- Hayashi T. Chapt. 25. In: Jacobsen EN, Pfaltz A, Yamamoto H, editors. Comprehensive Asymmetric Catalysis. Vol. II. Heidelberg: Springer Verlag; 1999.
- Blystone SL. Chem. Rev. 1989;89:1663–1679.
-
-
The trivial case of cross-coupling of chiral subunits where the newly formed carbon–carbon bond is not a stereogenic unit are excluded.
-
-
-
By far the largest category of transition metal-catalyzed, carbon-carbon and carbon-heteroatom bond forming reactions involves the asymmetric capture of π-allyl-metals derived from palladium, molybdenum, rhodium and iridium with a wide range of nucleophiles. These powerful reactions have been thoroughly reviewed and therefore will not be categorized here, see: Lu Z, Ma S. Angew. Chem., Int. Ed. 2008;47:258–297. Trost BM, Crawley ML. Chem. Rev. 2003:2921–2943. Pfaltz A, Lautens M. Chapt. 24. In: Jacobsen EN, Pfaltz A, Yamamoto H, editors. Comprehensive Asymmetric Catalysis. Vol. II. Heidelberg: Springer Verlag; 1999.
-
-
-
For a definition of internal and external selection see: Denmark SE, Almstead NG. Allylation of Carbonyls: Methodology and Stereochemistry. In: Otera J, editor. Modern Carbonyl Chemistry. Weinheim: Wiley–VCH; 2000. pp. 299–402.
-
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

