Neural correlates of variations in event processing during learning in basolateral amygdala
- PMID: 20164330
- PMCID: PMC2838173
- DOI: 10.1523/JNEUROSCI.5781-09.2010
Neural correlates of variations in event processing during learning in basolateral amygdala
Abstract
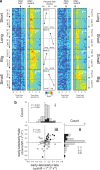
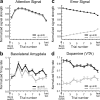
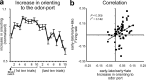
The discovery that dopamine neurons signal errors in reward prediction has demonstrated that concepts empirically derived from the study of animal behavior can be used to understand the neural implementation of reward learning. Yet the learning theory models linked to phasic dopamine activity treat attention to events such as cues and rewards as static quantities; other models, such as Pearce-Hall, propose that learning might be influenced by variations in processing of these events. A key feature of these accounts is that event processing is modulated by unsigned rather than signed reward prediction errors. Here we tested whether neural activity in rat basolateral amygdala conforms to this pattern by recording single units in a behavioral task in which rewards were unexpectedly delivered or omitted. We report that neural activity at the time of reward is providing an unsigned error signal with characteristics consistent with those postulated by these models. This neural signal increased immediately after a change in reward, and stronger firing was evident whether the value of the reward increased or decreased. Further, as predicted by these models, the change in firing developed over several trials as expectations for reward were repeatedly violated. This neural signal was correlated with faster orienting to predictive cues after changes in reward, and abolition of the signal by inactivation of basolateral amygdala disrupted this change in orienting and retarded learning in response to changes in reward. These results suggest that basolateral amygdala serves a critical function in attention for learning.
Figures

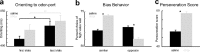
Similar articles
-
Dopamine neurons encode the better option in rats deciding between differently delayed or sized rewards.Nat Neurosci. 2007 Dec;10(12):1615-24. doi: 10.1038/nn2013. Epub 2007 Nov 18. Nat Neurosci. 2007. PMID: 18026098 Free PMC article.
-
Attention-related Pearce-Kaye-Hall signals in basolateral amygdala require the midbrain dopaminergic system.Biol Psychiatry. 2012 Dec 15;72(12):1012-9. doi: 10.1016/j.biopsych.2012.05.023. Epub 2012 Jul 3. Biol Psychiatry. 2012. PMID: 22763185 Free PMC article.
-
Normal aging alters learning and attention-related teaching signals in basolateral amygdala.J Neurosci. 2012 Sep 19;32(38):13137-44. doi: 10.1523/JNEUROSCI.2393-12.2012. J Neurosci. 2012. PMID: 22993430 Free PMC article.
-
Predictive reward signal of dopamine neurons.J Neurophysiol. 1998 Jul;80(1):1-27. doi: 10.1152/jn.1998.80.1.1. J Neurophysiol. 1998. PMID: 9658025 Review.
-
Involvement of basal ganglia and orbitofrontal cortex in goal-directed behavior.Prog Brain Res. 2000;126:193-215. doi: 10.1016/S0079-6123(00)26015-9. Prog Brain Res. 2000. PMID: 11105648 Review.
Cited by
-
Aversive-bias and stage-selectivity in neurons of the primate amygdala during acquisition, extinction, and overnight retention.J Neurosci. 2012 Jun 20;32(25):8598-610. doi: 10.1523/JNEUROSCI.0323-12.2012. J Neurosci. 2012. PMID: 22723701 Free PMC article.
-
Change detection, multiple controllers, and dynamic environments: insights from the brain.J Exp Anal Behav. 2013 Jan;99(1):74-84. doi: 10.1002/jeab.5. Epub 2012 Dec 5. J Exp Anal Behav. 2013. PMID: 23344989 Free PMC article.
-
Neural substrates of cognitive biases during probabilistic inference.Nat Commun. 2016 Apr 26;7:11393. doi: 10.1038/ncomms11393. Nat Commun. 2016. PMID: 27116102 Free PMC article.
-
Optogenetic Dissection of Temporal Dynamics of Amygdala-Striatal Interplay during Risk/Reward Decision Making.eNeuro. 2018 Dec 10;5(6):ENEURO.0422-18.2018. doi: 10.1523/ENEURO.0422-18.2018. eCollection 2018 Nov-Dec. eNeuro. 2018. PMID: 30627636 Free PMC article.
-
Functional connectivity between the amygdala and prefrontal cortex underlies processing of emotion ambiguity.Transl Psychiatry. 2023 Oct 28;13(1):334. doi: 10.1038/s41398-023-02625-w. Transl Psychiatry. 2023. PMID: 37898626 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
- R01-MH053667/MH/NIMH NIH HHS/United States
- R01-AG027097/AG/NIA NIH HHS/United States
- R01-DA015718/DA/NIDA NIH HHS/United States
- R37 MH053667/MH/NIMH NIH HHS/United States
- R01 MH053667/MH/NIMH NIH HHS/United States
- K01 DA021609/DA/NIDA NIH HHS/United States
- F31 MH080514/MH/NIMH NIH HHS/United States
- T32-NS07375/NS/NINDS NIH HHS/United States
- R01 DA015718/DA/NIDA NIH HHS/United States
- K01DA021609/DA/NIDA NIH HHS/United States
- T32 NS007375/NS/NINDS NIH HHS/United States
- R01 AG027097/AG/NIA NIH HHS/United States
- F31-MH080514/MH/NIMH NIH HHS/United States
LinkOut - more resources
Full Text Sources
