Cilia organize ependymal planar polarity
- PMID: 20164345
- PMCID: PMC2873868
- DOI: 10.1523/JNEUROSCI.3744-09.2010
Cilia organize ependymal planar polarity
Abstract
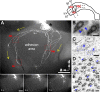
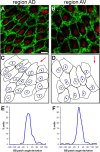
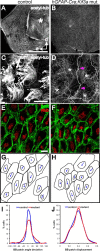
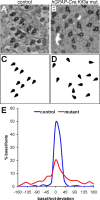
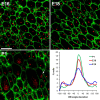
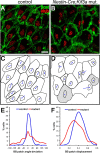
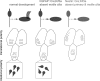
Multiciliated epithelial cells, called ependymal cells, line the ventricles in the adult brain. Most ependymal cells are born prenatally and are derived from radial glia. Ependymal cells have a remarkable planar polarization that determines orientation of ciliary beating and propulsion of CSF. Disruption of ependymal ciliary beating, by injury or disease, results in aberrant CSF circulation and hydrocephalus, a common disorder of the CNS. Very little is known about the mechanisms guiding ependymal planar polarity and whether this organization is acquired during ependymal cell development or is already present in radial glia. Here we show that basal bodies in ependymal cells in the lateral ventricle walls of adult mice are polarized in two ways: (1) rotational; angle of individual basal bodies with respect to their long axis and (2) translational; the position of basal bodies on the apical surface of the cell. Conditional ablation of motile cilia disrupted rotational orientation, but translational polarity was largely preserved. In contrast, translational polarity was dramatically affected when radial glial primary cilia were ablated earlier in development. Remarkably, radial glia in the embryo have a translational polarity that predicts the orientation of mature ependymal cells. These results suggest that ependymal planar cell polarity is a multistep process initially organized by primary cilia in radial glia and then refined by motile cilia in ependymal cells.
Figures
References
-
- al-Shroof M, Karnik AM, Karnik AA, Longshore J, Sliman NA, Khan FA. Ciliary dyskinesia associated with hydrocephalus and mental retardation in a Jordanian family. Mayo Clin Proc. 2001;76:1219–1224. - PubMed
-
- Banizs B, Pike MM, Millican CL, Ferguson WB, Komlosi P, Sheetz J, Bell PD, Schwiebert EM, Yoder BK. Dysfunctional cilia lead to altered ependyma and choroid plexus function, and result in the formation of hydrocephalus. Development. 2005;132:5329–5339. - PubMed
-
- Boisvieux-Ulrich E, Laine MC, Sandoz D. The orientation of ciliary basal bodies in quail oviduct is related to the ciliary beating cycle commencement. Biol Cell. 1985;55:147–150. - PubMed
-
- Bruni JE. Ependymal development, proliferation, and functions: a review. Microsc Res Tech. 1998;41:2–13. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
