Homeostatic regulation of synaptic excitability: tonic GABA(A) receptor currents replace I(h) in cortical pyramidal neurons of HCN1 knock-out mice
- PMID: 20164346
- PMCID: PMC2830721
- DOI: 10.1523/JNEUROSCI.3771-09.2010
Homeostatic regulation of synaptic excitability: tonic GABA(A) receptor currents replace I(h) in cortical pyramidal neurons of HCN1 knock-out mice
Abstract
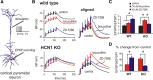
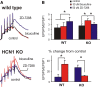
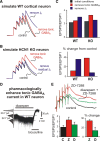
Homeostatic control of synaptic efficacy is often mediated by dynamic regulation of excitatory synaptic receptors. Here, we report a novel form of homeostatic synaptic plasticity based on regulation of shunt currents that control dendritosomatic information transfer. In cortical pyramidal neurons from wild-type mice, HCN1 channels underlie a dendritic hyperpolarization-activated cationic current (I(h)) that serves to limit temporal summation of synaptic inputs. In HCN1 knock-out mice, as expected, I(h) is reduced in pyramidal neurons and its effects on synaptic summation are strongly diminished. Unexpectedly, we found a markedly enhanced bicuculline- and L-655,708-sensitive background GABA(A) current in these cells that could be attributed to selective upregulation of GABA(A) alpha5 subunit expression in the cortex of HCN1 knock-out mice. Strikingly, despite diminished I(h), baseline sublinear summation of evoked EPSPs was unchanged in pyramidal neurons from HCN1 knock-out mice; however, blocking tonic GABA(A) currents with bicuculline enhanced synaptic summation more strongly in pyramidal cells from HCN1 knock-out mice than in those cells from wild-type mice. Increasing tonic GABA(A) receptor conductance in the context of reduced I(h), using computational or pharmacological approaches, restored normal baseline synaptic summation, as observed in neurons from HCN1 knock-out mice. These data indicate that upregulation of alpha5 subunit-mediated GABA(A) receptor tonic current compensates quantitatively for loss of dendritic I(h) in cortical pyramidal neurons from HCN1 knock-out mice to maintain normal synaptic summation; they further imply that dendritosomatic synaptic efficacy is a controlled variable for homeostatic regulation of cortical neuron excitability in vivo.
Figures




References
-
- Biel M, Wahl-Schott C, Michalakis S, Zong X. Hyperpolarization-activated cation channels: from genes to function. Physiol Rev. 2009;89:847–885. - PubMed
-
- Bonin RP, Martin LJ, MacDonald JF, Orser BA. α5GABAA receptors regulate the intrinsic excitability of mouse hippocampal pyramidal neurons. J Neurophysiol. 2007;98:2244–2254. - PubMed
-
- Brickley SG, Revilla V, Cull-Candy SG, Wisden W, Farrant M. Adaptive regulation of neuronal excitability by a voltage-independent potassium conductance. Nature. 2001;409:88–92. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
