Amyloid beta induces the morphological neurodegenerative triad of spine loss, dendritic simplification, and neuritic dystrophies through calcineurin activation
- PMID: 20164348
- PMCID: PMC2841957
- DOI: 10.1523/JNEUROSCI.4456-09.2010
Amyloid beta induces the morphological neurodegenerative triad of spine loss, dendritic simplification, and neuritic dystrophies through calcineurin activation
Abstract
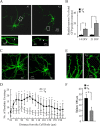
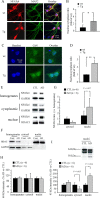
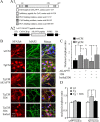
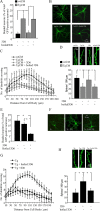
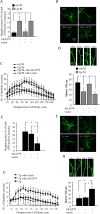
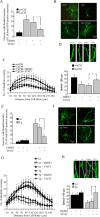
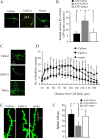
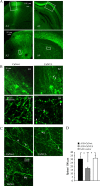
Amyloid beta (Abeta)-containing plaques are surrounded by dystrophic neurites in the Alzheimer's disease (AD) brain, but whether and how plaques induce these neuritic abnormalities remain unknown. We tested the hypothesis that soluble oligomeric assemblies of Abeta, which surround plaques, induce calcium-mediated secondary cascades that lead to dystrophic changes in local neurites. We show that soluble Abeta oligomers lead to activation of the calcium-dependent phosphatase calcineurin (CaN) (PP2B), which in turn activates the transcriptional factor nuclear factor of activated T cells (NFAT). Activation of these signaling pathways, even in the absence of Abeta, is sufficient to produce a virtual phenocopy of Abeta-induced dystrophic neurites, dendritic simplification, and dendritic spine loss in both neurons in culture and in the adult mouse brain. Importantly, the morphological deficits in the vicinity of Abeta deposits in a mouse model of AD are ameliorated by CaN inhibition, supporting the hypothesis that CaN-NFAT are aberrantly activated by Abeta and that CaN-NFAT activation is responsible for disruption of neuronal structure near plaques. In accord with this, we also detect increased levels of an active form of CaN and NFATc4 in the nuclear fraction from the cortex of patients with AD. Thus, Abeta appears to mediate the neurodegeneration of AD, at least in part, by activation of CaN and subsequent NFAT-mediated downstream cascades.
Figures
References
-
- Agostinho P, Lopes JP, Velez Z, Oliveira CR. Overactivation of calcineurin induced by amyloid-beta and prion proteins. Neurochem Int. 2008;52:1226–1233. - PubMed
-
- Aramburu J, Yaffe MB, López-Rodríguez C, Cantley LC, Hogan PG, Rao A. Affinity-driven peptide selection of an NFAT inhibitor more selective than cyclosporin A. Science. 1999;285:2129–2133. - PubMed
-
- Berridge MJ, Lipp P, Bootman MD. The versatility and universality of calcium signalling. Nat Rev Mol Cell Biol. 2000;1:11–21. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous
