Head-free gaze shifts provide further insights into the role of the medial cerebellum in the control of primate saccadic eye movements
- PMID: 20164388
- PMCID: PMC2853288
- DOI: 10.1152/jn.91361.2008
Head-free gaze shifts provide further insights into the role of the medial cerebellum in the control of primate saccadic eye movements
Abstract
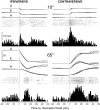
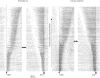
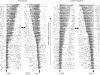
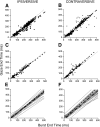
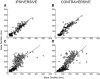
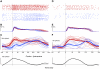
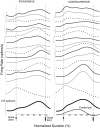
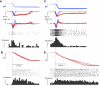
This study examines how signals generated in the oculomotor cerebellum could be involved in the control of gaze shifts, which rapidly redirect the eyes from one object to another. Neurons in the caudal fastigial nucleus (cFN), the output of the oculomotor cerebellum, discharged when monkeys made horizontal head-unrestrained gaze shifts, composed of an eye saccade and a head movement. Eighty-seven percent of our neurons discharged a burst of spikes for both ipsiversive and contraversive gaze shifts. In both directions, burst end was much better timed with gaze end than was burst start with gaze start, was well correlated with eye end, and was poorly correlated with head end or the time of peak head velocity. Moreover, bursts accompanied all head-unrestrained gaze shifts whether the head moved or not. Therefore we conclude that the cFN is not part of the pathway that controls head movement. For contraversive gaze shifts, the early part of the burst was correlated with gaze acceleration. Thereafter, the burst of the neuronal population continued throughout the prolonged deceleration of large gaze shifts. For a majority of neurons, gaze duration was correlated with burst duration; for some, gaze amplitude was less well correlated with the number of spikes. Therefore we suggest that the population burst provides an acceleration boost for high acceleration (smaller) contraversive gaze shifts and helps maintain the drive required to extend the deceleration of large contraversive gaze shifts. In contrast, the ipsiversive population burst, which is less well correlated with gaze metrics but whose peak rate occurs before gaze end, seems responsible primarily for terminating the gaze shift.
Figures
References
-
- Batton R, Jayaraman A, Ruggiero D, Carpenter M. Fastigial efferent projections in the monkey: an autoradiographic study. J Comp Neurol 174: 281–306, 1977 - PubMed
-
- Becker W. Metrics. In: The Neurobiology of Saccadic Eye Movements, edited by Wurtz B, Goldberg M. Amsterdam: Elsevier, 1989, p. 13–67 - PubMed
-
- Brettler S, Fuchs A. Activity of caudal fastigial nucleus neurons during head-unrestrained gaze shifts in rhesus monkeys. Soc Neurosci Abstr 27: 405.11, 2001
-
- Brettler S, Fuchs A, Ling L. Discharge patterns of cerebellar output neurons in the caudal fastigial nucleus during head-free gaze shifts in primates. Ann NY Acad Sci 1004: 61–68, 2003 - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

