The estrous cycle of the ewe is resistant to disruption by repeated, acute psychosocial stress
- PMID: 20164438
- PMCID: PMC2874503
- DOI: 10.1095/biolreprod.109.078774
The estrous cycle of the ewe is resistant to disruption by repeated, acute psychosocial stress
Abstract
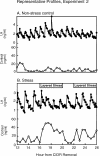
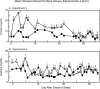
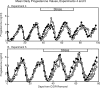
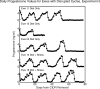
Five experiments were conducted to test the hypothesis that psychosocial stress interferes with the estrous cycle of sheep. In experiment 1, ewes were repeatedly isolated during the follicular phase. Timing, amplitude, and duration of the preovulatory luteinizing hormone (LH) surge were not affected. In experiment 2, follicular-phase ewes were subjected twice to a "layered stress" paradigm consisting of sequential, hourly application of isolation, restraint, blindfold, and predator cues. This reduced the LH pulse amplitude but did not affect the LH surge. In experiment 3, different acute stressors were given sequentially within the follicular phase: food denial plus unfamiliar noises and forced exercise, layered stress, exercise around midnight, and transportation. This, too, did not affect the LH surge. In experiment 4, variable acute psychosocial stress was given every 1-2 days for two entire estrous cycles; this did not disrupt any parameter of the cycle monitored. Lastly, experiment 5 examined whether the psychosocial stress paradigms of experiment 4 would disrupt the cycle and estrous behavior if sheep were metabolically stressed by chronic food restriction. Thirty percent of the food-restricted ewes exhibited deterioration of estrous cycle parameters followed by cessation of cycles and failure to express estrous behavior. However, disruption was not more evident in ewes that also encountered psychosocial stress. Collectively, these findings indicate the estrous cycle of sheep is remarkably resistant to disruption by acute bouts of psychosocial stress applied intermittently during either a single follicular phase or repeatedly over two estrous cycles.
Figures
Similar articles
-
Exposure to Acute Psychosocial Stress Disrupts the Luteinizing Hormone Surge Independent of Estrous Cycle Alterations in Female Mice.Endocrinology. 2017 Aug 1;158(8):2593-2602. doi: 10.1210/en.2017-00341. Endocrinology. 2017. PMID: 28549157 Free PMC article.
-
LH pulse frequency and the emergence and growth of ovarian antral follicular waves in the ewe during the luteal phase of the estrous cycle.Reprod Biol Endocrinol. 2009 Jul 28;7:78. doi: 10.1186/1477-7827-7-78. Reprod Biol Endocrinol. 2009. PMID: 19638235 Free PMC article.
-
Progesterone priming is essential for the full expression of the positive feedback effect of estradiol in inducing the preovulatory gonadotropin-releasing hormone surge in the ewe.Endocrinology. 1999 Jan;140(1):165-70. doi: 10.1210/endo.140.1.6444. Endocrinology. 1999. PMID: 9886822
-
Suppression of the secretion of luteinizing hormone due to isolation/restraint stress in gonadectomised rams and ewes is influenced by sex steroids.J Endocrinol. 1999 Mar;160(3):469-81. doi: 10.1677/joe.0.1600469. J Endocrinol. 1999. PMID: 10076193
-
A role for estradiol in enhancing luteinizing hormone pulse frequency during the follicular phase of the estrous cycle of sheep.Endocrinology. 1983 Oct;113(4):1333-9. doi: 10.1210/endo-113-4-1333. Endocrinology. 1983. PMID: 6684548
Cited by
-
Regulation of the gonadotropin-releasing hormone neuron during stress.J Neuroendocrinol. 2022 May;34(5):e13098. doi: 10.1111/jne.13098. Epub 2022 Feb 6. J Neuroendocrinol. 2022. PMID: 35128742 Free PMC article. Review.
-
Exposure to Acute Psychosocial Stress Disrupts the Luteinizing Hormone Surge Independent of Estrous Cycle Alterations in Female Mice.Endocrinology. 2017 Aug 1;158(8):2593-2602. doi: 10.1210/en.2017-00341. Endocrinology. 2017. PMID: 28549157 Free PMC article.
-
Faecal glucocorticoid metabolites and body temperature in Australian merino ewes (Ovis aries) during summer artificial insemination (AI) program.PLoS One. 2018 Jan 30;13(1):e0191961. doi: 10.1371/journal.pone.0191961. eCollection 2018. PLoS One. 2018. PMID: 29381759 Free PMC article.
-
Intermittent fasting dietary restriction regimen negatively influences reproduction in young rats: a study of hypothalamo-hypophysial-gonadal axis.PLoS One. 2013;8(1):e52416. doi: 10.1371/journal.pone.0052416. Epub 2013 Jan 29. PLoS One. 2013. PMID: 23382817 Free PMC article.
-
Influence of stress-induced intermediates on gonadotropin gene expression in gonadotrope cells.Mol Cell Endocrinol. 2014 Mar 25;385(1-2):71-7. doi: 10.1016/j.mce.2013.08.014. Epub 2013 Sep 4. Mol Cell Endocrinol. 2014. PMID: 24012628 Free PMC article. Review.
References
-
- Rivier C, Rivest S.Effect of stress on the activity of the hypothalamic-pituitary-gonadal axis: peripheral and central mechanisms. Biol Reprod 1991; 45: 523–532. - PubMed
-
- Ferin M.Neuropeptides, the stress response, and the hypothalamo-pituitary-gonadal axis in the female rhesus monkey. Ann N Y Acad Sci 1993; 697: 106–116. - PubMed
-
- Dobson H, Smith RF.What is stress, and how does it affect reproduction? Anim Reprod Sci 2000; 60–61: 743–752. - PubMed
-
- Tilbrook AJ, Turner AI, Clarke IJ.Effects of stress on reproduction in nonrodent mammals: the role of glucocorticoids and sex differences. Rev Reprod 2000; 5: 105–113. - PubMed
-
- Breen KM, Karsch FJ.New insights regarding glucocorticoids, stress and gonadotropin suppression. Front Neuroendocrinol 2006; 27: 233–245. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

