Lentiviral vectors incorporating a human elongation factor 1alpha promoter for the treatment of canine leukocyte adhesion deficiency
- PMID: 20164857
- PMCID: PMC3461956
- DOI: 10.1038/gt.2010.7
Lentiviral vectors incorporating a human elongation factor 1alpha promoter for the treatment of canine leukocyte adhesion deficiency
Abstract
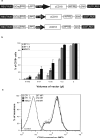
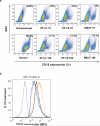
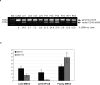
Canine leukocyte adhesion deficiency (CLAD) provides a unique large animal model for testing new therapeutic approaches for the treatment of children with leukocyte adhesion deficiency (LAD). In our CLAD model, we examined two different fragments of the human elongation factor 1alpha (EF1alpha) promoter (EF1alphaL, 1189 bp and EF1alphaS, 233 bp) driving the expression of canine CD18 in a self-inactivating (SIN) lentiviral vector. The EF1alphaS vector resulted in the highest levels of canine CD18 expression in CLAD CD34(+) cells in vitro. Subsequently, autologous CD34(+) bone marrow cells from four CLAD pups were transduced with the EF1alphaS vector and infused following a non-myeloablative dose of 200 cGy total-body irradiation. None of the CLAD pups achieved levels of circulating CD18(+) neutrophils sufficient to reverse the CLAD phenotype, and all four animals were euthanized because of infections within 9 weeks of treatment. These results indicate that the EF1alphaS promoter-driven CD18 expression in the context of a RRLSIN lentiviral vector does not lead to sufficient numbers of CD18(+) neutrophils in vivo to reverse the CLAD phenotype when used in a non-myeloablative transplant regimen in dogs.
Figures
Similar articles
-
Gene therapy of canine leukocyte adhesion deficiency using lentiviral vectors with human CD11b and CD18 promoters driving canine CD18 expression.Mol Ther. 2011 Jan;19(1):113-21. doi: 10.1038/mt.2010.203. Epub 2010 Sep 21. Mol Ther. 2011. PMID: 20859258 Free PMC article.
-
Gene therapy for canine leukocyte adhesion deficiency with lentiviral vectors using the murine stem cell virus and human phosphoglycerate kinase promoters.Hum Gene Ther. 2011 Jun;22(6):689-96. doi: 10.1089/hum.2010.130. Epub 2011 Apr 11. Hum Gene Ther. 2011. PMID: 21275758 Free PMC article.
-
Efficiency of different fragment lengths of the ubiquitous chromatin opening element HNRPA2B1-CBX3 in driving human CD18 gene expression within self-inactivating lentiviral vectors for gene therapy applications.Gene. 2019 Aug 20;710:265-272. doi: 10.1016/j.gene.2019.06.016. Epub 2019 Jun 11. Gene. 2019. PMID: 31200085
-
Leukocyte adhesion deficiency in children and Irish setter dogs.Pediatr Res. 2004 Mar;55(3):363-7. doi: 10.1203/01.PDR.0000111287.74989.1B. Epub 2004 Jan 7. Pediatr Res. 2004. PMID: 14711903 Review.
-
Gene therapy for leukocyte adhesion deficiency.Curr Opin Mol Ther. 2000 Aug;2(4):383-8. Curr Opin Mol Ther. 2000. PMID: 11249768 Review.
Cited by
-
Gene therapy of canine leukocyte adhesion deficiency using lentiviral vectors with human CD11b and CD18 promoters driving canine CD18 expression.Mol Ther. 2011 Jan;19(1):113-21. doi: 10.1038/mt.2010.203. Epub 2010 Sep 21. Mol Ther. 2011. PMID: 20859258 Free PMC article.
-
Engineering Next-Generation BET-Independent MLV Vectors for Safer Gene Therapy.Mol Ther Nucleic Acids. 2017 Jun 16;7:231-245. doi: 10.1016/j.omtn.2017.04.002. Epub 2017 Apr 12. Mol Ther Nucleic Acids. 2017. PMID: 28624199 Free PMC article.
-
Plaque associated microglia hyper-secrete extracellular vesicles and accelerate tau propagation in a humanized APP mouse model.Mol Neurodegener. 2021 Mar 22;16(1):18. doi: 10.1186/s13024-021-00440-9. Mol Neurodegener. 2021. PMID: 33752701 Free PMC article.
-
Gene transfer into hematopoietic stem cells as treatment for primary immunodeficiency diseases.Int J Hematol. 2014 Apr;99(4):383-92. doi: 10.1007/s12185-014-1524-z. Epub 2014 Feb 1. Int J Hematol. 2014. PMID: 24488786 Review.
-
Recent insights into the pathobiology of innate immune deficiencies.Curr Allergy Asthma Rep. 2011 Oct;11(5):369-77. doi: 10.1007/s11882-011-0212-9. Curr Allergy Asthma Rep. 2011. PMID: 21814768 Free PMC article. Review.
References
-
- Trowald-Wigh G, Hakansson L, Johannisson A, Norrgren L, Hard af Segerstad C. Leucocyte adhesion protein deficiency in Irish setter dogs. Vet Immunol Immunopathol. 1992;32:261–280. - PubMed
-
- Creevy KE, Bauer TR, Jr., Tuschong LM, Embree LJ, Colenda L, Cogan K, et al. Canine leukocyte adhesion deficiency colony for investigation of novel hematopoietic therapies. Vet Immunol Immunopathol. 2003;94:11–22. - PubMed
-
- Kijas JM, Bauer TR, Jr., Gafvert S, Marklund S, Trowald-Wigh G, Johannisson A, et al. A missense mutation in the β2 integrin gene (ITGB2) causes canine leukocyte adhesion deficiency. Genomics. 1999;61:101–107. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

