The cyclic nucleotide-gated ion channel CNGA3 contributes to coolness-induced responses of Grueneberg ganglion neurons
- PMID: 20165899
- PMCID: PMC11115782
- DOI: 10.1007/s00018-010-0296-8
The cyclic nucleotide-gated ion channel CNGA3 contributes to coolness-induced responses of Grueneberg ganglion neurons
Abstract
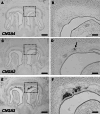
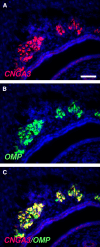
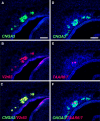
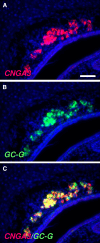
Localized to the vestibule of the nasal cavity, neurons of the Grueneberg ganglion (GG) respond to cool ambient temperatures. The molecular mechanisms underlying this thermal response are still elusive. Recently, it has been suggested that cool temperatures may activate a cyclic guanosine monophosphate (cGMP) pathway in the GG, which would be reminiscent of thermosensory neurons in Caenorhabditis elegans. In search for other elements of such a cascade, we have found that the cyclic nucleotide-gated ion channel CNGA3 was strongly expressed in the GG and that expression of CNGA3 was confined to those cells that are responsive to coolness. Further experiments revealed that the response of GG neurons to cool temperatures was significantly reduced in CNGA3-deficient mice compared to wild-type conspecifics. The observation that a cGMP-activated non-selective cation channel significantly contributes to the coolness-evoked response in GG neurons strongly suggests that a cGMP cascade is part of the transduction process.
Figures


Similar articles
-
The Grueneberg ganglion: signal transduction and coding in an olfactory and thermosensory organ involved in the detection of alarm pheromones and predator-secreted kairomones.Cell Tissue Res. 2021 Jan;383(1):535-548. doi: 10.1007/s00441-020-03380-w. Epub 2021 Jan 6. Cell Tissue Res. 2021. PMID: 33404842 Review.
-
Chemo- and thermosensory responsiveness of Grueneberg ganglion neurons relies on cyclic guanosine monophosphate signaling elements.Neurosignals. 2011;19(4):198-209. doi: 10.1159/000329333. Epub 2011 Aug 31. Neurosignals. 2011. PMID: 21893936
-
Expression of cGMP signaling elements in the Grueneberg ganglion.Histochem Cell Biol. 2009 Jan;131(1):75-88. doi: 10.1007/s00418-008-0514-8. Epub 2008 Oct 1. Histochem Cell Biol. 2009. PMID: 18830617
-
Grueneberg ganglion neurons are finely tuned cold sensors.J Neurosci. 2010 Jun 2;30(22):7563-8. doi: 10.1523/JNEUROSCI.0608-10.2010. J Neurosci. 2010. PMID: 20519530 Free PMC article.
-
The Grueneberg ganglion: a novel sensory system in the nose.Histol Histopathol. 2010 Jul;25(7):909-15. doi: 10.14670/HH-25.909. Histol Histopathol. 2010. PMID: 20503179 Review.
Cited by
-
Ion channels of cold transduction and transmission.J Gen Physiol. 2024 Oct 7;156(10):e202313529. doi: 10.1085/jgp.202313529. Epub 2024 Jul 25. J Gen Physiol. 2024. PMID: 39051992 Free PMC article. Review.
-
Integrating temperature with odor processing in the olfactory bulb.J Neurosci. 2015 May 20;35(20):7892-902. doi: 10.1523/JNEUROSCI.0571-15.2015. J Neurosci. 2015. PMID: 25995474 Free PMC article.
-
The Grueneberg ganglion: signal transduction and coding in an olfactory and thermosensory organ involved in the detection of alarm pheromones and predator-secreted kairomones.Cell Tissue Res. 2021 Jan;383(1):535-548. doi: 10.1007/s00441-020-03380-w. Epub 2021 Jan 6. Cell Tissue Res. 2021. PMID: 33404842 Review.
-
Electrophysiological characterization of Grueneberg ganglion olfactory neurons: spontaneous firing, sodium conductance, and hyperpolarization-activated currents.J Neurophysiol. 2012 Sep;108(5):1318-34. doi: 10.1152/jn.00907.2011. Epub 2012 May 30. J Neurophysiol. 2012. PMID: 22649209 Free PMC article.
-
The thermosensitive potassium channel TREK-1 contributes to coolness-evoked responses of Grueneberg ganglion neurons.Cell Mol Neurobiol. 2014 Jan;34(1):113-22. doi: 10.1007/s10571-013-9992-x. Epub 2013 Oct 8. Cell Mol Neurobiol. 2014. PMID: 24101433 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

