Osteoblastic induction on calcium phosphate cement-chitosan constructs for bone tissue engineering
- PMID: 20166217
- PMCID: PMC2875315
- DOI: 10.1002/jbm.a.32665
Osteoblastic induction on calcium phosphate cement-chitosan constructs for bone tissue engineering
Abstract
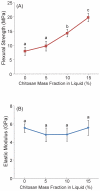
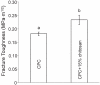
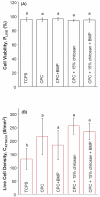
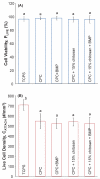
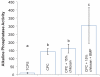
Calcium phosphate cement (CPC) is osteoconductive and moldable, and it can conform to complex cavity shapes and set in situ to form hydroxyapatite. Chitosan could increase the strength and toughness of CPC, but there has been no investigation on recombinant human bone morphogenic protein-2 (rhBMP-2) delivery via CPC-chitosan composite and its effect on osteogenic induction of cells. The objective of this research was to investigate the mechanical properties and osteoblastic induction of MC3T3-E1 cells cultured on CPC-containing chitosan and rhBMP-2. Cell viability for CPC with chitosan and rhBMP-2 was comparable with that of control CPC, whereas the CPC-chitosan composite was stronger and tougher than CPC control. After 14 days, osteoblastic induction was quantified by measuring alkaline phosphatase (ALP) activity. ALP (mean +/- SD; n = 6) of cells seeded on conventional CPC without rhBMP-2 was (143 +/- 19) (mM pNpp/min)/(mug DNA). The addition of chitosan resulted in an ALP of 161 +/- 27. Further addition of rhBMP-2 to the CPC-chitosan composite increased the ALP to 305 +/- 111 (p < 0.05). All ALP activity on CPC composites was significantly higher when compared with the 10.0 +/- 3.3 of tissue culture polystyrene (p < 0.05). Flexural strength of CPC containing 15% (mass fraction) chitosan was 19.8 +/- 1.4 MPa, which is more than double the 8.0 +/- 1.4 MPa of conventional CPC (p < 0.05). The addition of chitosan to CPC increased the fracture toughness from 0.18 +/- 0.01 MPa.m(1/2) to 0.23 +/- 0.02 MPa.m(1/2) (p < 0.05). The relatively high strength, self-hardening CPC-chitosan composite scaffold is promising as a moderate load-bearing matrix for bone repair, with potential to serve as an injectable delivery vehicle for osteoinductive growth factors to promote osteoblastic induction and bone regeneration. (c) 2010 Wiley Periodicals, Inc. J Biomed Mater Res, 2010.
Figures


Similar articles
-
Culture human mesenchymal stem cells with calcium phosphate cement scaffolds for bone repair.J Biomed Mater Res B Appl Biomater. 2010 Apr;93(1):93-105. doi: 10.1002/jbm.b.31563. J Biomed Mater Res B Appl Biomater. 2010. PMID: 20091907 Free PMC article.
-
Injectable and strong nano-apatite scaffolds for cell/growth factor delivery and bone regeneration.Dent Mater. 2008 Sep;24(9):1212-22. doi: 10.1016/j.dental.2008.02.001. Epub 2008 Mar 21. Dent Mater. 2008. PMID: 18359072 Free PMC article.
-
Synergistic reinforcement of in situ hardening calcium phosphate composite scaffold for bone tissue engineering.Biomaterials. 2004 Mar;25(6):1029-37. doi: 10.1016/s0142-9612(03)00608-2. Biomaterials. 2004. PMID: 14615168
-
Mesenchymal stem cell proliferation and differentiation on an injectable calcium phosphate-chitosan composite scaffold.Biomaterials. 2009 May;30(14):2675-82. doi: 10.1016/j.biomaterials.2009.01.022. Epub 2009 Feb 1. Biomaterials. 2009. PMID: 19187958 Free PMC article.
-
Delivering MC3T3-E1 cells into injectable calcium phosphate cement through alginate-chitosan microcapsules for bone tissue engineering.J Zhejiang Univ Sci B. 2014 Apr;15(4):382-92. doi: 10.1631/jzus.B1300132. J Zhejiang Univ Sci B. 2014. PMID: 24711359 Free PMC article.
Cited by
-
Combined construction of injectable tissue-engineered bone with CPC and BMMSCs and its study of repair effect on rabbit bone defect model.J Musculoskelet Neuronal Interact. 2020 Mar 3;20(1):142-148. J Musculoskelet Neuronal Interact. 2020. PMID: 32131379 Free PMC article.
-
Novel Calcium Phosphate Cement with Metformin-Loaded Chitosan for Odontogenic Differentiation of Human Dental Pulp Cells.Stem Cells Int. 2018 Nov 27;2018:7173481. doi: 10.1155/2018/7173481. eCollection 2018. Stem Cells Int. 2018. PMID: 30598667 Free PMC article.
-
Local delivery of alendronate eluting chitosan scaffold can effectively increase osteoblast functions and inhibit osteoclast differentiation.J Mater Sci Mater Med. 2012 Nov;23(11):2739-49. doi: 10.1007/s10856-012-4729-9. Epub 2012 Aug 1. J Mater Sci Mater Med. 2012. PMID: 22850978
-
The effect of mineral coating morphology on mesenchymal stem cell attachment and expansion.J Mater Chem. 2012 Dec 28;22(48):25288-25295. doi: 10.1039/C2JM33354F. J Mater Chem. 2012. PMID: 25663752 Free PMC article.
-
Human periodontal ligament stem cells on calcium phosphate scaffold delivering platelet lysate to enhance bone regeneration.RSC Adv. 2019 Dec 13;9(70):41161-41172. doi: 10.1039/c9ra08336g. eCollection 2019 Dec 9. RSC Adv. 2019. PMID: 35540034 Free PMC article.
References
-
- Salgado AJ, Coutinho OP, Reis RL. Bone tissue engineering: state of the art and future trends. Macromol Biosci. 2004;4:743–765. - PubMed
-
- Laurencin CT, Ambrosio AM, Borden MD, Cooper JA., Jr. Tissue engineering: orthopedic applications. Annu Rev Biomed Eng. 1999;1:19–46. 19-46. - PubMed
-
- Mackay AM, Beck SC, Murphy JM, Barry FP, Chichester CO, Pittenger MF. Chondrogenic differentiation of cultured human mesenchymal stem cells from marrow. Tissue Eng. 1998;4:415–428. - PubMed
-
- Winn SR, Uludag H, Hollinger JO. Carrier systems for bone morphogenetic proteins. Clin Orthop Relat Res. 1999:S95–106. - PubMed
-
- Reddi AH. Symbiosis of biotechnology and biomaterials: applications in tissue engineering of bone and cartilage. J Cell Biochem. 1994;56:192–195. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

