Cetuximab (erbitux)
- PMID: 20167650
- PMCID: PMC7964212
- DOI: 10.3174/ajnr.A2054
Cetuximab (erbitux)
Abstract
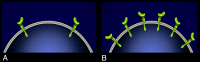
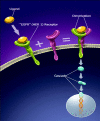
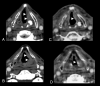
Cetuximab, a recombinant chimeric monoclonal antibody, has been successfully used in the treatment of the head and neck and colorectal cancers. We present a review of its mechanism of action, indications, side effects and economic issues, accompanied by a clinical example from our institution.
Figures

References
-
- Blick SK, Scott LJ. Cetuximab: a review of its use in squamous cell carcinoma of the head and neck and metastatic colorectal cancer. Drugs 2007; 67: 2585–607 - PubMed
-
- Wong SF. Cetuximab: an epidermal growth factor receptor monoclonal antibody for the treatment of colorectal cancer. Clin Ther 2005; 27: 684–94 - PubMed
-
- Steiner P, Joynes C, Bassi R, et al. . Tumor growth inhibition with cetuximab and chemotherapy in non-small cell lung cancer xenografts expressing wild-type and mutated epidermal growth factor receptor. Clin Cancer Res 2007; 13: 1540–51 - PubMed
-
- Erbitux (Cetuximab); [package insert]. New York and Princeton, New Jersey: ImClone Systems and Bristol-Myers Squibb; 2009
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous
