Dysbindin promotes the post-endocytic sorting of G protein-coupled receptors to lysosomes
- PMID: 20174469
- PMCID: PMC2824829
- DOI: 10.1371/journal.pone.0009325
Dysbindin promotes the post-endocytic sorting of G protein-coupled receptors to lysosomes
Abstract
Background: Dysbindin, a cytoplasmic protein long known to function in the biogenesis of specialized lysosome-related organelles (LROs), has been reported to reduce surface expression of D2 dopamine receptors in neurons. Dysbindin is broadly expressed, and dopamine receptors are members of the large family of G protein-coupled receptors (GPCRs) that function in diverse cell types. Thus we asked if dysbindin regulates receptor number in non-neural cells, and further investigated the cellular basis of this regulation.
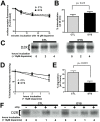
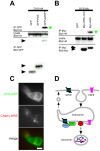
Methodology/principal findings: We used RNA interference to deplete endogenous dysbindin in HEK293 and HeLa cells, then used immunochemical and biochemical methods to assess expression and endocytic trafficking of epitope-tagged GPCRs. Dysbindin knockdown up-regulated surface expression of D2 receptors compared to D1 receptors, as reported previously in neurons. This regulation was not mediated by a change in D2 receptor endocytosis. Instead, dysbindin knockdown specifically reduced the subsequent trafficking of internalized D2 receptors to lysosomes. This distinct post-endocytic sorting function explained the minimal effect of dysbindin depletion on D1 receptors, which recycle efficiently and traverse the lysosomal pathway to only a small degree. Moreover, dysbindin regulated the delta opioid receptor, a more distantly related GPCR that is also sorted to lysosomes after endocytosis. Dysbindin was not required for lysosomal trafficking of all signaling receptors, however, as its depletion did not detectably affect down-regulation of the EGF receptor tyrosine kinase. Dysbindin co-immunoprecipitated with GASP-1 (or GPRASP-1), a cytoplasmic protein shown previously to modulate lysosomal trafficking of D2 dopamine and delta opioid receptors by direct interaction, and with HRS that is a core component of the conserved ESCRT machinery mediating lysosome biogenesis and sorting.
Conclusions/significance: These results identify a distinct, and potentially widespread function of dysbindin in promoting the sorting of specific GPCRs to lysosomes after endocytosis.
Conflict of interest statement
Figures



Similar articles
-
Role of mammalian vacuolar protein-sorting proteins in endocytic trafficking of a non-ubiquitinated G protein-coupled receptor to lysosomes.J Biol Chem. 2004 May 21;279(21):22522-31. doi: 10.1074/jbc.M311062200. Epub 2004 Mar 15. J Biol Chem. 2004. PMID: 15024011
-
Gαs regulates the post-endocytic sorting of G protein-coupled receptors.Nat Commun. 2014 Aug 4;5:4556. doi: 10.1038/ncomms5556. Nat Commun. 2014. PMID: 25089012 Free PMC article.
-
Ubiquitination regulates proteolytic processing of G protein-coupled receptors after their sorting to lysosomes.J Biol Chem. 2009 Jul 17;284(29):19361-70. doi: 10.1074/jbc.M109.001644. Epub 2009 May 11. J Biol Chem. 2009. PMID: 19433584 Free PMC article.
-
Atypical regulation of G protein-coupled receptor intracellular trafficking by ubiquitination.Curr Opin Cell Biol. 2014 Apr;27:44-50. doi: 10.1016/j.ceb.2013.11.004. Epub 2013 Dec 7. Curr Opin Cell Biol. 2014. PMID: 24680429 Free PMC article. Review.
-
Regulation of GPCRs by endocytic membrane trafficking and its potential implications.Annu Rev Pharmacol Toxicol. 2008;48:537-68. doi: 10.1146/annurev.pharmtox.48.113006.094830. Annu Rev Pharmacol Toxicol. 2008. PMID: 18184106 Review.
Cited by
-
Modulating neuromodulation by receptor membrane traffic in the endocytic pathway.Neuron. 2012 Oct 4;76(1):22-32. doi: 10.1016/j.neuron.2012.09.022. Neuron. 2012. PMID: 23040804 Free PMC article. Review.
-
Retinal biomarkers and pharmacological targets for Hermansky-Pudlak syndrome 7.Sci Rep. 2020 Mar 4;10(1):3972. doi: 10.1038/s41598-020-60931-5. Sci Rep. 2020. PMID: 32132582 Free PMC article.
-
The effect of clozapine on mRNA expression for genes encoding G protein-coupled receptors and the protein components of clathrin-mediated endocytosis.Psychiatr Genet. 2013 Aug;23(4):153-62. doi: 10.1097/YPG.0b013e32835fe51d. Psychiatr Genet. 2013. PMID: 23811784 Free PMC article.
-
Endo-lysosomal sorting of G-protein-coupled receptors by ubiquitin: Diverse pathways for G-protein-coupled receptor destruction and beyond.Traffic. 2019 Feb;20(2):101-109. doi: 10.1111/tra.12619. Epub 2018 Nov 18. Traffic. 2019. PMID: 30353650 Free PMC article. Review.
-
Apoptotic signalling targets the post-endocytic sorting machinery of the death receptor Fas/CD95.Nat Commun. 2019 Jul 15;10(1):3105. doi: 10.1038/s41467-019-11025-y. Nat Commun. 2019. PMID: 31308371 Free PMC article.
References
-
- Pierce KL, Lefkowitz RJ. Classical and new roles of beta-arrestins in the regulation of G-protein-coupled receptors. Nat Rev Neurosci. 2001;2:727–733. - PubMed
-
- Gainetdinov RR, Premont RT, Bohn LM, Lefkowitz RJ, Caron MG. Desensitization of G protein-coupled receptors and neuronal functions. Annu Rev Neurosci. 2004;27:107–144. - PubMed
-
- Sawa A, Snyder SH. Schizophrenia: diverse approaches to a complex disease. Science. 2002;296:692–695. - PubMed
-
- Benson MA, Newey SE, Martin-Rendon E, Hawkes R, Blake DJ. Dysbindin, a novel coiled-coil-containing protein that interacts with the dystrobrevins in muscle and brain. J Biol Chem. 2001;276:24232–24241. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

