Nevirapine-associated early hepatotoxicity: incidence, risk factors, and associated mortality in a primary care ART programme in South Africa
- PMID: 20174653
- PMCID: PMC2822855
- DOI: 10.1371/journal.pone.0009183
Nevirapine-associated early hepatotoxicity: incidence, risk factors, and associated mortality in a primary care ART programme in South Africa
Abstract
Background: The majority of antiretroviral treatment programmes in sub-Saharan Africa are scaling up antiretroviral treatment using a fixed dose first-line antiretroviral regimen containing stavudine, lamivudine, and nevirapine. One of the primary concerns with the use of this regimen is nevirapine-associated hepatotoxicity.
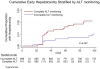
Methodology/principal findings: Study participants were 1809 HIV-infected, antiretroviral naïve adults initiating nevirapine-based antiretroviral therapy between November 2002 and December 2006. The primary outcome was early hepatotoxicity. Secondary outcomes were associations with hepatotoxicity and mortality at six months. The cumulative proportion of early hepatotoxicity ranged from 1.0-2.0% giving an incidence-rate at 102 days of 3.6-7.6 per 100 person-years. Median time to hepatotoxicity was 32 (IQR 28-58) days. At 12 weeks, only 8% of patients had alanine aminotransferase monitoring at all the time-points recommended by national guidelines. No association was found between age, gender, baseline CD4 count, concurrent tuberculosis infection, prior participation in a prevention of mother-to-child-transmission program, or baseline weight and early hepatotoxicity. There was no association between early hepatotoxicity and mortality.
Conclusions: The cumulative proportion of early hepatotoxicity in nevirapine based antiretroviral therapy was low in this resource-constrained setting. Hepatotoxicity was not associated with mortality. Frequent routine monitoring of alanine aminotransferase proved difficult to implement in this public sector primary care programme. Focused monitoring in the first month may be a more cost-effective and pragmatic option in settings with limited resources. Correlation with clinical signs and symptoms may allow future alanine aminotransferase testing to be dictated by clinical criteria.
Conflict of interest statement
Figures
References
-
- (2003) Scaling Up Antiretroviral Therapy in Resource-Limited Settings. Treatment Guidelines for a Public Health Approach. Geneva. 2003 - PubMed
-
- Forna F, Liechty CA, Solberg P, Asiimwe F, Were W, et al. Clinical toxicity of highly active antiretroviral therapy in a home-based AIDS care program in rural Uganda. J Acquir Immune Defic Syndr. 2007;44:456–462. - PubMed
-
- Pharmaceuticals BI. Ridgefield, CT.: 2005. Viramune Package Insert.
-
- Ridgefield, CT: Boehringer Ingelheim Pharmaceuticals; 2008. Viramune Package Insert.
-
- Martinez E, Blanco JL, Arnaiz JA, Perez-Cuevas JB, Mocroft A, et al. Hepatotoxicity in HIV-1-infected patients receiving nevirapine-containing antiretroviral therapy. AIDS. 2001;15:1261–1268. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials