In vivo bioluminescence tomography with a blocking-off finite-difference SP3 method and MRI/CT coregistration
- PMID: 20175496
- PMCID: PMC2803718
- DOI: 10.1118/1.3273034
In vivo bioluminescence tomography with a blocking-off finite-difference SP3 method and MRI/CT coregistration
Abstract
Purpose: Bioluminescence imaging is a research tool for studying gene expression levels in small animal models of human disease. Bioluminescence light, however, is strongly scattered in biological tissue and no direct image of the light-emitting reporter probe's location can be obtained. Therefore, the authors have developed a linear image reconstruction method for bioluminescence tomography (BLT) that recovers the three-dimensional spatial bioluminescent source distribution in small animals.
Methods: The proposed reconstruction method uses third-order simplified spherical harmonics (SP3) solutions to the equation of radiative transfer for modeling the bioluminescence light propagation in optically nonuniform tissue. The SP3 equations and boundary conditions are solved with a finite-difference (FD) technique on a regular grid. The curved geometry of the animal surface was taken into account with a blocking-off region method for regular grids. Coregistered computed tomography (CT) and magnetic resonance (MR) images provide information regarding the geometry of the skin surface and internal organs. The inverse source problem is defined as an algebraic system of linear equations for the unknown source distribution and is iteratively solved given multiview and multispectral boundary measurements. The average tissue absorption parameters, which are used for the image reconstruction process, were calculated with an evolution strategy (ES) from in vivo measurements using an implanted pointlike source of known location and spectrum. Moreover, anatomical information regarding the location of the internal organs and other tissue structures within the animal's body are provided by coregistered MR images.
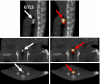
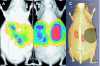
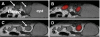
Results: First, the authors recovered the wavelength-dependent absorption coefficients (average error of 14%) with the ES under ideal conditions by using a numerical mouse model. Next, they reconstructed the average absorption coefficient of a small animal by using an artificial implanted light source and the validated ES. Last, they conducted two in vivo animal experiments and recovered the spatial location of the implanted light source and the spatial distribution of a bioluminescent reporter system located in the kidneys. The source reconstruction results were coregistered to CT and MR images. They further found that accurate bioluminescence image reconstructions could be obtained when segmenting a voidlike cyst with low-scattering and absorption parameters, whereas inaccurate image reconstructions were obtained when assuming a uniform optical parameter distribution instead. The image reconstructions were completed within 23 min on a 3 GHz Intel processor.
Conclusions: The authors demonstrated on in vivo examples that the combination of anatomical coregistration, accurate optical tissue properties, multispectral acquisition, and a blocking-off FD-SP3 solution of the radiative transfer model significantly improves the accuracy of the BLT reconstructions.
Figures


Similar articles
-
Bioluminescence tomography with CT/MRI co-registration.Annu Int Conf IEEE Eng Med Biol Soc. 2009;2009:6327-30. doi: 10.1109/IEMBS.2009.5333170. Annu Int Conf IEEE Eng Med Biol Soc. 2009. PMID: 19964154
-
An adaptive regularization parameter choice strategy for multispectral bioluminescence tomography.Med Phys. 2011 Nov;38(11):5933-44. doi: 10.1118/1.3635221. Med Phys. 2011. PMID: 22047358
-
Registration of planar bioluminescence to magnetic resonance and x-ray computed tomography images as a platform for the development of bioluminescence tomography reconstruction algorithms.J Biomed Opt. 2009 Mar-Apr;14(2):024045. doi: 10.1117/1.3120495. J Biomed Opt. 2009. PMID: 19405773 Free PMC article.
-
Development of volume conductor and source models to localize epileptic foci.J Clin Neurophysiol. 2007 Apr;24(2):101-19. doi: 10.1097/WNP.0b013e318038fb3e. J Clin Neurophysiol. 2007. PMID: 17414966 Review.
-
Non-invasive three-dimensional imaging of Escherichia coli K1 infection using diffuse light imaging tomography combined with micro-computed tomography.Methods. 2017 Aug 15;127:62-68. doi: 10.1016/j.ymeth.2017.05.005. Epub 2017 May 15. Methods. 2017. PMID: 28522324 Review.
Cited by
-
Making the brain glow: in vivo bioluminescence imaging to study neurodegeneration.Mol Neurobiol. 2013 Jun;47(3):868-82. doi: 10.1007/s12035-012-8379-1. Epub 2012 Nov 29. Mol Neurobiol. 2013. PMID: 23192390 Review.
-
Efficient numerical modelling of time-domain light propagation in curved 3D absorbing and scattering media with finite differences.Biomed Opt Express. 2021 Feb 16;12(3):1422-1436. doi: 10.1364/BOE.413854. eCollection 2021 Mar 1. Biomed Opt Express. 2021. PMID: 33796363 Free PMC article.
-
Adaptive shrinking reconstruction framework for cone-beam X-ray luminescence computed tomography.Biomed Opt Express. 2020 Jun 12;11(7):3717-3732. doi: 10.1364/BOE.393970. eCollection 2020 Jul 1. Biomed Opt Express. 2020. PMID: 33014562 Free PMC article.
-
Bioluminescence Tomography-Guided Radiation Therapy for Preclinical Research.Int J Radiat Oncol Biol Phys. 2016 Apr 1;94(5):1144-53. doi: 10.1016/j.ijrobp.2015.11.039. Epub 2015 Dec 14. Int J Radiat Oncol Biol Phys. 2016. PMID: 26876954 Free PMC article.
-
Multi-modal molecular diffuse optical tomography system for small animal imaging.Meas Sci Technol. 2013;24(10):105405. doi: 10.1088/0957-0233/24/10/105405. Meas Sci Technol. 2013. PMID: 24954977 Free PMC article.
References
-
- Blasberg R. G., “Molecular imaging and cancer,” Molecular Cancer Therapeutics 2, 335–343 (2003). - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

