Low-dose dexamethasone prevents endotoxaemia-induced muscle protein loss and impairment of carbohydrate oxidation in rat skeletal muscle
- PMID: 20176631
- PMCID: PMC2872737
- DOI: 10.1113/jphysiol.2009.183699
Low-dose dexamethasone prevents endotoxaemia-induced muscle protein loss and impairment of carbohydrate oxidation in rat skeletal muscle
Abstract
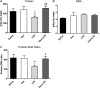
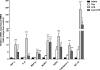
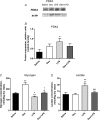
We recently provided evidence suggesting a role for cytokine-mediated inhibition of Akt/Forkhead box O 1 (FOXO1) signalling in the induction of muscle atrophy and impairment of muscle carbohydrate oxidation during lipopolysaccharide (LPS)-induced endotoxaemia in rats. We hypothesized that a low-dose dexamethasone (Dex; anti-inflammatory agent) infusion during endotoxaemia would prevent the LPS-induced impairment of Akt/FOXO1 signalling, and therefore prevent the muscle atrophy and impairment of carbohydrate oxidation. Chronically instrumented Sprague-Dawley rats received a continuous intravenous infusion of LPS (15 microg kg(-1) h(-1)), Dex (12.5 microg kg(-1) h(-1)), Dex+LPS or saline for 24 h at 0.4 ml h(-1). LPS infusion caused haemodynamic changes consistent with a hyperdynamic circulation and induced increases in muscle tumour necrosis factor-alpha (TNF-alpha; 10-fold, P < 0.001), interleukin-6 (IL-6; 14-fold, P < 0.001) and metallothionein-1A (MT-1A; 187-fold, P < 0.001) mRNA expression. Dex co-administration abolished most of the haemodynamic effects of LPS and reduced the increase in muscle TNF-alpha, IL-6 and MT-1A by 51% (P < 0.01), 85% (P < 0.001) and 58% (P < 0.01), respectively. Dex infusion during endotoxaemia also prevented the LPS-induced 40% reduction in the muscle protein:DNA ratio and decrease in Akt phosphorylation, and partially prevented the reduction in FOXO1 phosphorylation. However, Dex did not prevent the LPS-mediated increase in muscle atrophy F-box (MAFbx) and muscle RING finger 1 (MuRF1) mRNA expression, but did significantly reduce the LPS-mediated increase in cathepsin-L mRNA expression and enzyme activity by 43% (P < 0.001) and 53% (P < 0.05), respectively. Furthermore, Dex suppressed LPS-induced pyruvate dehydrogenase kinase 4 (PDK4) mRNA upregulation by approximately 50% (P < 0.01), and prevented LPS-mediated muscle glycogen breakdown and lactate accumulation. Thus, low-dose Dex infusion during endotoxaemia prevented muscle atrophy and the impairment of carbohydrate oxidation, potentially through suppression of cytokine-mediated Akt/FOXO inhibition, and blunting of cathepsin-L-mediated lysosomal protein breakdown.
Figures

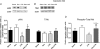
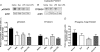
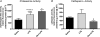
 and
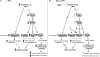
and  arrows indicate upregulation/increased activity and downregulation/decreased activity, respectively, and arrow thickness represents the magnitude of upregulation/increased activity and downregulation/decreased activity.
arrows indicate upregulation/increased activity and downregulation/decreased activity, respectively, and arrow thickness represents the magnitude of upregulation/increased activity and downregulation/decreased activity.Comment in
-
Out-FOX(O)ing proteolysis in sepsis.J Physiol. 2010 Apr 15;588(Pt 8):1193. doi: 10.1113/jphysiol.2010.189498. J Physiol. 2010. PMID: 20395471 Free PMC article. No abstract available.
Similar articles
-
A potential role for Akt/FOXO signalling in both protein loss and the impairment of muscle carbohydrate oxidation during sepsis in rodent skeletal muscle.J Physiol. 2008 Nov 15;586(22):5589-600. doi: 10.1113/jphysiol.2008.160150. Epub 2008 Sep 25. J Physiol. 2008. PMID: 18818241 Free PMC article.
-
Out-FOX(O)ing proteolysis in sepsis.J Physiol. 2010 Apr 15;588(Pt 8):1193. doi: 10.1113/jphysiol.2010.189498. J Physiol. 2010. PMID: 20395471 Free PMC article. No abstract available.
-
Peroxisome proliferator-activated receptor γ agonism attenuates endotoxaemia-induced muscle protein loss and lactate accumulation in rats.Clin Sci (Lond). 2017 Jun 22;131(13):1437-1447. doi: 10.1042/CS20170958. Print 2017 Jul 1. Clin Sci (Lond). 2017. PMID: 28536293
-
Gut-derived low-grade endotoxaemia, atherothrombosis and cardiovascular disease.Nat Rev Cardiol. 2023 Jan;20(1):24-37. doi: 10.1038/s41569-022-00737-2. Epub 2022 Jul 15. Nat Rev Cardiol. 2023. PMID: 35840742 Free PMC article. Review.
-
The impact of immobilisation and inflammation on the regulation of muscle mass and insulin resistance: different routes to similar end-points.J Physiol. 2019 Mar;597(5):1259-1270. doi: 10.1113/JP275444. Epub 2018 Aug 18. J Physiol. 2019. PMID: 29968251 Free PMC article. Review.
Cited by
-
Structure to function: muscle failure in critically ill patients.J Physiol. 2010 Dec 1;588(Pt 23):4641-8. doi: 10.1113/jphysiol.2010.197632. Epub 2010 Oct 20. J Physiol. 2010. PMID: 20961998 Free PMC article. Review.
-
The Effects of Glutamine Supplementation on Liver Inflammatory Response and Protein Metabolism in Muscle of Lipopolysaccharide-Challenged Broilers.Animals (Basel). 2024 Feb 1;14(3):480. doi: 10.3390/ani14030480. Animals (Basel). 2024. PMID: 38338123 Free PMC article.
-
Control of skeletal muscle atrophy in response to disuse: clinical/preclinical contentions and fallacies of evidence.Am J Physiol Endocrinol Metab. 2016 Sep 1;311(3):E594-604. doi: 10.1152/ajpendo.00257.2016. Epub 2016 Jul 5. Am J Physiol Endocrinol Metab. 2016. PMID: 27382036 Free PMC article. Review.
-
Skeletal muscle dysregulation in rheumatoid arthritis: Metabolic and molecular markers in a rodent model and patients.PLoS One. 2020 Jul 7;15(7):e0235702. doi: 10.1371/journal.pone.0235702. eCollection 2020. PLoS One. 2020. PMID: 32634159 Free PMC article.
-
A membrane glucocorticoid receptor mediates the rapid/non-genomic actions of glucocorticoids in mammalian skeletal muscle fibres.J Physiol. 2013 Oct 15;591(20):5171-85. doi: 10.1113/jphysiol.2013.256586. Epub 2013 Jul 22. J Physiol. 2013. PMID: 23878367 Free PMC article.
References
-
- Angus DC, Linde-Zwirble WT, Lidicker J, Clermont G, Carcillo J, Pinsky MR. Epidemiology of severe sepsis in the United States: analysis of incidence, outcome, and associated costs of care. Crit Care Med. 2001;29:1303–1310. - PubMed
-
- Annane D. Corticosteroids for septic shock. Crit Care Med. 2001;29:S117–120. - PubMed
-
- Bergmeyer HU. Methods in Enzymatic Analysis. 3rd edn. V. John Wiley & Sons; 1983. pp. 207–209.
-
- Blough E, Dineen B, Esser K. Extraction of nuclear proteins from striated muscle tissue. Biotechniques. 1999;26:202–204. 206. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

