Characterization of the colchicine binding site on avian tubulin isotype betaVI
- PMID: 20178367
- PMCID: PMC2852430
- DOI: 10.1021/bi100159p
Characterization of the colchicine binding site on avian tubulin isotype betaVI
Abstract
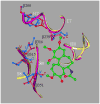
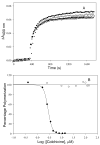
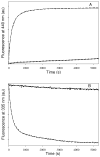
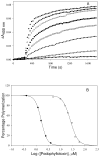
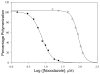
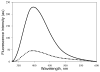
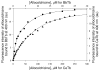
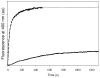
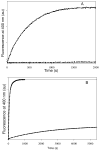
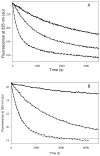
Tubulin, the basic component of microtubules, is present in most eukaryotic cells as multiple gene products, called isotypes. The major tubulin isotypes are highly conserved in terms of structure and drug binding capabilities. Tubulin isotype betaVI, however, is significantly divergent from the other isotypes in sequence, assembly properties, and function. It is the major beta-tubulin isotype of hematopoietic tissue and forms the microtubules of platelet marginal bands. The interaction of the major tubulin isotypes betaI, betaII, betaIII, and betaIotaV with antimicrotubule drugs has been widely studied, but little is known about the drug binding properties of tubulin isotype betaVI. In this investigation, we characterize the activity of various colchicine site ligands with tubulin isolated from Gallus gallus erythrocytes (CeTb), which is approximately 95% betaVI. Colchicine binding is thought to be a universal property of higher eukaryotic tubulin; however, we were unable to detect colchicine binding to CeTb under any experimental conditions. Podophyllotoxin and nocodazole, other colchicine site ligands with divergent structures, were able to inhibit paclitaxel-induced CeTb assembly. Surprisingly, the colchicine isomer allocolchicine also inhibited CeTb assembly and displayed measurable, moderate affinity for CeTb (K(a) = 0.18 x 10(5) M(-1) vs 5.0 x 10(5) M(-1) for bovine brain tubulin). Since allocolchicine and colchicine differ in their C ring structures, the two C ring colchicine analogues were also tested for CeTb binding. Kinetic experiments indicate that thiocolchicine and chlorocolchicine bind to CeTb, but very slowly and with low affinity. Molecular modeling of CeTb identified five divergent amino acid residues within 6 A of the colchicine binding site compared to betaI, betaII, and betaIV; three of these amino acids are also altered in betaIII-tubulin. Interestingly, the altered amino acids are in the vicinity of the A ring region of the colchicine binding site rather than the C ring region. We propose that the amino acid differences in the binding site constrict the A ring binding domain in CeTb, which interferes with the positioning of the trimethoxyphenyl A ring and prevents C ring binding site interactions from efficiently occurring. Allocolchicine is able to accommodate the altered binding mode because of its smaller ring size and more flexible C ring substituents. The sequence of the colchicine binding domain of CeTb isotype betaVI is almost identical to that of its human hematopoietic counterpart. Thus, through analysis of the interactions of ligands with CeTb, it may be possible to discover colchicine site ligands that specifically target tubulin in human hematopoietic cells.
Figures

References
-
- Morejohn LC, Fosket DE. The biochemistry of compounds with anti-microtubule activity in plant-cells. Pharmacol Ther. 1991;51:217–230. - PubMed
-
- Kohler P. The biochemical basis of anthelmintic action and resistance. Int J Parasitol. 2001;31:336–345. - PubMed
-
- Jordan A, Hadfield JA, Lawrence NJ, McGown AT. Tubulin as a target for anticancer drugs: Agents which interact with the mitotic spindle. Med Res Rev. 1998;18:259–296. - PubMed
-
- Luduena RF. Multiple forms of tubulin: Different gene products and covalent modifications, in. International Review of Cytology - a Survey of Cell Biology. 1998;178:207–275. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

