The anti-tumorigenic properties of peroxisomal proliferator-activated receptor alpha are arachidonic acid epoxygenase-mediated
- PMID: 20178979
- PMCID: PMC2857132
- DOI: 10.1074/jbc.M109.081554
The anti-tumorigenic properties of peroxisomal proliferator-activated receptor alpha are arachidonic acid epoxygenase-mediated
Abstract
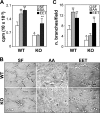
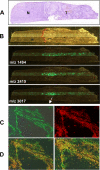
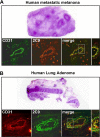
Prevalence and mortality make cancer a health challenge in need of effective and better tolerated therapeutic approaches, with tumor angiogenesis identified as a promising target for drug development. The epoxygenase products, the epoxyeicosatrienoic acids, are pro-angiogenic, and down-regulation of their biosynthesis by peroxisomal proliferator-activated receptor alpha (PPARalpha) ligands reduces tumor angiogenesis and growth. Endothelial cells lacking a Cyp2c44 epoxygenase, a PPARalpha target, show reduced proliferative and tubulogenic activities that are reversed by the enzyme's metabolites. In a mouse xenograft model of tumorigenesis, disruption of the host Cyp2c44 gene causes marked reductions in tumor volume, mass, and vascularization. The relevance of these studies to human cancer is indicated by the demonstration that: (a) activation of human PPARalpha down-regulates endothelial cell CYP2C9 epoxygenase expression and blunts proliferation and tubulogenesis, (b) in a PPARalpha-humanized mouse model, activation of the receptor inhibits tumor angiogenesis and growth, and (c) the CYP2C9 epoxygenase is expressed in the vasculature of human tumors. The identification of anti-angiogenic/anti-tumorigenic properties of PPARalpha points to a role for the receptor and its epoxygenase regulatory target in the pathophysiology of cancer, and for its ligands as candidates for the development of a new generation of safer and better tolerated anti-cancer drugs.
Figures




Similar articles
-
Peroxisomal proliferator-activated receptor-alpha-dependent inhibition of endothelial cell proliferation and tumorigenesis.J Biol Chem. 2007 Jun 15;282(24):17685-95. doi: 10.1074/jbc.M701429200. Epub 2007 Apr 3. J Biol Chem. 2007. PMID: 17405874
-
PPARα ligand, AVE8134, and cyclooxygenase inhibitor therapy synergistically suppress lung cancer growth and metastasis.BMC Cancer. 2019 Dec 2;19(1):1166. doi: 10.1186/s12885-019-6379-5. BMC Cancer. 2019. PMID: 31791289 Free PMC article.
-
PPARα activation can help prevent and treat non-small cell lung cancer.Cancer Res. 2014 Jan 15;74(2):621-31. doi: 10.1158/0008-5472.CAN-13-1928. Epub 2013 Dec 3. Cancer Res. 2014. PMID: 24302581 Free PMC article.
-
Hydrogen peroxide generation in peroxisome proliferator-induced oncogenesis.Mutat Res. 2000 Mar 17;448(2):159-77. doi: 10.1016/s0027-5107(99)00234-1. Mutat Res. 2000. PMID: 10725470 Review.
-
The role and regulation of the peroxisome proliferator activated receptor alpha in human liver.Biochimie. 2017 May;136:75-84. doi: 10.1016/j.biochi.2016.12.019. Epub 2017 Jan 8. Biochimie. 2017. PMID: 28077274 Review.
Cited by
-
Epoxyeicosatrienoic acids: a double-edged sword in cardiovascular diseases and cancer.J Clin Invest. 2012 Jan;122(1):19-22. doi: 10.1172/JCI61453. Epub 2011 Dec 19. J Clin Invest. 2012. PMID: 22182836 Free PMC article.
-
Cytochrome P450-derived epoxyeicosatrienoic acids accelerate wound epithelialization and neovascularization in the hairless mouse ear wound model.Langenbecks Arch Surg. 2011 Dec;396(8):1245-53. doi: 10.1007/s00423-011-0838-z. Epub 2011 Sep 2. Langenbecks Arch Surg. 2011. PMID: 21887579
-
Fatty Acids Metabolism: The Bridge Between Ferroptosis and Ionizing Radiation.Front Cell Dev Biol. 2021 Jun 24;9:675617. doi: 10.3389/fcell.2021.675617. eCollection 2021. Front Cell Dev Biol. 2021. PMID: 34249928 Free PMC article. Review.
-
Genetic variation in the eicosanoid pathway is associated with non-small-cell lung cancer (NSCLC) survival.PLoS One. 2017 Jul 13;12(7):e0180471. doi: 10.1371/journal.pone.0180471. eCollection 2017. PLoS One. 2017. PMID: 28704416 Free PMC article.
-
The arachidonic acid monooxygenase: from biochemical curiosity to physiological/pathophysiological significance.J Lipid Res. 2018 Nov;59(11):2047-2062. doi: 10.1194/jlr.R087882. Epub 2018 Aug 28. J Lipid Res. 2018. PMID: 30154230 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

