Targeted deletion of hepatocyte ABCA1 leads to very low density lipoprotein triglyceride overproduction and low density lipoprotein hypercatabolism
- PMID: 20178985
- PMCID: PMC2852959
- DOI: 10.1074/jbc.M109.096933
Targeted deletion of hepatocyte ABCA1 leads to very low density lipoprotein triglyceride overproduction and low density lipoprotein hypercatabolism
Abstract
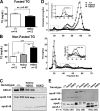
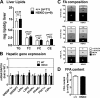
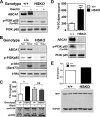
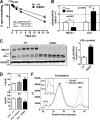
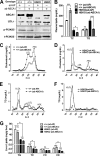
Loss of ABCA1 activity in Tangier disease (TD) is associated with abnormal apoB lipoprotein (Lp) metabolism in addition to the complete absence of high density lipoprotein (HDL). We used hepatocyte-specific ABCA1 knock-out (HSKO) mice to test the hypothesis that hepatic ABCA1 plays dual roles in regulating Lp metabolism and nascent HDL formation. HSKO mice recapitulated the TD lipid phenotype with postprandial hypertriglyceridemia, markedly decreased LDL, and near absence of HDL. Triglyceride (TG) secretion was 2-fold higher in HSKO compared with wild type mice, primarily due to secretion of larger TG-enriched VLDL secondary to reduced hepatic phosphatidylinositol 3-kinase signaling. HSKO mice also displayed delayed clearance of postprandial TG and reduced post-heparin plasma lipolytic activity. In addition, hepatic LDLr expression and plasma LDL catabolism were increased 2-fold in HSKO compared with wild type mice. Last, adenoviral repletion of hepatic ABCA1 in HSKO mice normalized plasma VLDL TG and hepatic phosphatidylinositol 3-kinase signaling, with a partial recovery of HDL cholesterol levels, providing evidence that hepatic ABCA1 is involved in the reciprocal regulation of apoB Lp production and HDL formation. These findings suggest that altered apoB Lp metabolism in TD subjects may result from hepatic VLDL TG overproduction and increased hepatic LDLr expression and highlight hepatic ABCA1 as an important regulatory factor for apoB-containing Lp metabolism.
Figures

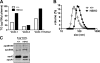
References
-
- Wellington C. L., Walker E. K., Suarez A., Kwok A., Bissada N., Singaraja R., Yang Y. Z., Zhang L. H., James E., Wilson J. E., Francone O., McManus B. M., Hayden M. R. (2002) Lab. Invest. 82, 273–283 - PubMed
-
- Assman G., von Eckardstein A., Brewer H. B., Jr. (2001) in The Metabolic and Molecular Bases of Inherited Disease (Scriver C. R., Beaudet A. L., Sly W. S., Valle D., Childs B., Kinzler K. W., Volkman B. F. eds) pp. 2937–2960, McGraw-Hill, New York
-
- Bodzioch M., Orsó E., Klucken J., Langmann T., Böttcher A., Diederich W., Drobnik W., Barlage S., Büchler C., Porsch-Ozcürümez M., Kaminski W. E., Hahmann H. W., Oette K., Rothe G., Aslanidis C., Lackner K. J., Schmitz G. (1999) Nat. Genet. 22, 347–351 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

