Systemic immune suppression in glioblastoma: the interplay between CD14+HLA-DRlo/neg monocytes, tumor factors, and dexamethasone
- PMID: 20179016
- PMCID: PMC2940665
- DOI: 10.1093/neuonc/noq001
Systemic immune suppression in glioblastoma: the interplay between CD14+HLA-DRlo/neg monocytes, tumor factors, and dexamethasone
Abstract
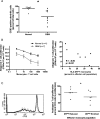
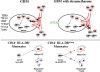
Patients with glioblastoma (GBM) exhibit profound systemic immune defects that affect the success of conventional and immune-based treatments. A better understanding of the contribution of the tumor and/or therapy on systemic immune suppression is necessary for improved therapies, to monitor negative effects of novel treatments, to improve patient outcomes, and to increase understanding of this complex system. To characterize the immune profile of GBM patients, we phenotyped peripheral blood and compared these to normal donors. In doing so, we identified changes in systemic immunity associated with both the tumor and dexamethasone treated tumor bearing patients. In particular, dexamethasone exacerbated tumor associated lymphopenia primarily in the T cell compartment. We have also identified unique tumor and dexamethasone dependent altered monocyte phenotypes. The major population of altered monocytes (CD14(+)HLA-DR(lo/neg)) had a phenotype distinct from classical myeloid suppressor cells. These cells inhibited T cell proliferation, were unable to fully differentiate into mature dendritic cells, were associated with dexamethasone-mediated changes in CCL2 levels, and could be re-created in vitro using tumor supernatants. We provide evidence that tumors express high levels of CCL2, can contain high numbers of CD14(+) cells, that tumor supernatants can transform CD14(+)HLA-DR(+) cells into CD14(+)HLA-DR(lo/neg) immune suppressors, and that dexamethasone reduces CCL2 in vitro and is correlated with reduction of CCL2 in vivo. Consequently, we have developed a model for tumor mediated systemic immune suppression via recruitment and transformation of CD14(+) cells.
Figures





References
-
- Stupp R, Mason WP, van den Bent MJ, et al. Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med. 2005;352:987–996. - PubMed
-
- Emens LA, Jaffe EM. Leveraging the activity of tumor vaccines with cytotoxic chemotherapy. Cancer Res. 2005;65:8059–8064. - PubMed
-
- Haynes NM, van der Most RG, Lake RA, Smyth MJ. Immunogenic anti-cancer chemotherapy as an emerging concept. Curr Opin Immunol. 2008;20:1–13. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

