Cytotoxic effects induced by docetaxel, gefitinib, and cyclopamine on side population and nonside population cell fractions from human invasive prostate cancer cells
- PMID: 20179163
- PMCID: PMC3228252
- DOI: 10.1158/1535-7163.MCT-09-1013
Cytotoxic effects induced by docetaxel, gefitinib, and cyclopamine on side population and nonside population cell fractions from human invasive prostate cancer cells
Abstract
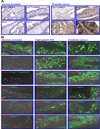
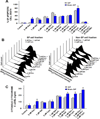
The present study has been undertaken to establish the therapeutic benefit of cotargeting epidermal growth factor receptor (EGFR) and sonic hedgehog pathways by using gefitinib and cyclopamine, respectively, for improving the efficacy of the current chemotherapeutic drug docetaxel to counteract the prostate cancer progression from locally invasive to metastatic and recurrent disease stages. The data from immuofluorescence analyses revealed that EGFR/Tyr(1173)-pEGFR, sonic hedgehog ligand, smoothened coreceptor, and GLI-1 were colocalized with the CD133(+) stem cell-like marker in a small subpopulation of prostate cancer cells. These signaling molecules were also present in the bulk tumor mass of CD133(-) prostate cancer cells with a luminal phenotype detected in patient's adenocarcinoma tissues. Importantly, the results revealed that the CD133(+)/CD44(high)/AR(-/low) side population (SP) cell fraction endowed with a high self-renewal potential isolated from tumorigenic and invasive WPE1-NB26 cells by the Hoechst dye technique was insensitive to the current chemotherapeutic drug, docetaxel. In contrast, the docetaxel treatment induced significant antiproliferative and apoptotic effects on the CD133(-)/CD44(low)/AR(+) non-SP cell fraction isolated from the WPE1-NB26 cell line. Of therapeutic interest, the results have also indicated that combined docetaxel, gefitinib, and cyclopamine induced greater antiproliferative and apoptotic effects on SP and non-SP cell fractions isolated from WPE1-NB26 cells than individual drugs or two-drug combinations. Altogether, these observations suggest that EGFR and sonic hedgehog cascades may represent the potential therapeutic targets of great clinical interest to eradicate the total prostate cancer cell mass and improve the current docetaxel-based therapies against locally advanced and invasive prostate cancers, and thereby prevent metastases and disease relapse.
Figures



Similar articles
-
Combined targeting of epidermal growth factor receptor and hedgehog signaling by gefitinib and cyclopamine cooperatively improves the cytotoxic effects of docetaxel on metastatic prostate cancer cells.Mol Cancer Ther. 2007 Mar;6(3):967-78. doi: 10.1158/1535-7163.MCT-06-0648. Mol Cancer Ther. 2007. PMID: 17363490
-
Pathobiological implications of the expression of EGFR, pAkt, NF-κB and MIC-1 in prostate cancer stem cells and their progenies.PLoS One. 2012;7(2):e31919. doi: 10.1371/journal.pone.0031919. Epub 2012 Feb 23. PLoS One. 2012. PMID: 22384099 Free PMC article.
-
Combined inhibition of epidermal growth factor receptor and cyclooxygenase-2 leads to greater anti-tumor activity of docetaxel in advanced prostate cancer.PLoS One. 2013 Oct 14;8(10):e76169. doi: 10.1371/journal.pone.0076169. eCollection 2013. PLoS One. 2013. PMID: 24155892 Free PMC article.
-
Animal models relevant to human prostate carcinogenesis underlining the critical implication of prostatic stem/progenitor cells.Biochim Biophys Acta. 2011 Aug;1816(1):25-37. doi: 10.1016/j.bbcan.2011.03.001. Epub 2011 Mar 17. Biochim Biophys Acta. 2011. PMID: 21396984 Free PMC article. Review.
-
New advances on critical implications of tumor- and metastasis-initiating cells in cancer progression, treatment resistance and disease recurrence.Histol Histopathol. 2010 Aug;25(8):1057-73. doi: 10.14670/HH-25.1057. Histol Histopathol. 2010. PMID: 20552555 Free PMC article. Review.
Cited by
-
Hypoxia-inducing factors as master regulators of stemness properties and altered metabolism of cancer- and metastasis-initiating cells.J Cell Mol Med. 2013 Jan;17(1):30-54. doi: 10.1111/jcmm.12004. Epub 2013 Jan 10. J Cell Mol Med. 2013. PMID: 23301832 Free PMC article.
-
Clinical Theragnostic Potential of Diverse miRNA Expressions in Prostate Cancer: A Systematic Review and Meta-Analysis.Cancers (Basel). 2020 May 9;12(5):1199. doi: 10.3390/cancers12051199. Cancers (Basel). 2020. PMID: 32397507 Free PMC article.
-
Therapeutic targeting of the prostate cancer microenvironment.Nat Rev Urol. 2010 Sep;7(9):494-509. doi: 10.1038/nrurol.2010.134. Nat Rev Urol. 2010. PMID: 20818327 Review.
-
HPMA copolymer-based combination therapy toxic to both prostate cancer stem/progenitor cells and differentiated cells induces durable anti-tumor effects.J Control Release. 2013 Dec 28;172(3):946-53. doi: 10.1016/j.jconrel.2013.09.005. Epub 2013 Sep 14. J Control Release. 2013. PMID: 24041709 Free PMC article.
-
Cancer Stem Cell Functions in Hepatocellular Carcinoma and Comprehensive Therapeutic Strategies.Cells. 2020 May 26;9(6):1331. doi: 10.3390/cells9061331. Cells. 2020. PMID: 32466488 Free PMC article. Review.
References
-
- Mimeault M, Batra SK. Recent advances on multiple tumorigenic cascades involved in prostatic cancer progression and targeting therapies. Carcinogenesis. 2006;27:1–22. - PubMed
-
- Litwin MS, Gore JL, Kwan L, et al. Quality of life after surgery, external beam irradiation, or brachytherapy for early-stage prostate cancer. Cancer. 2007;109:2239–2247. - PubMed
-
- Gignac GA, Morris MJ, Hussain M. Castration resistant, taxane naive metastatic prostate cancer: Current clinical approaches and future directions. J Urol. 2007;178:S30–S35. - PubMed
-
- Tannock IF, de Wit R, Berry WR, et al. Docetaxel plus prednisone or mitoxantrone plus prednisone for advanced prostate cancer. N Engl J Med. 2004;351:1502–1512. - PubMed
-
- Petrylak DP, Tangen CM, Hussain MH, et al. Docetaxel and estramustine compared with mitoxantrone and prednisone for advanced refractory prostate cancer. N Engl J Med. 2004;351:1513–1520. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

