Eph receptors and ephrins in cancer: bidirectional signalling and beyond
- PMID: 20179713
- PMCID: PMC2921274
- DOI: 10.1038/nrc2806
Eph receptors and ephrins in cancer: bidirectional signalling and beyond
Abstract
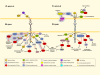
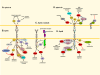
The Eph receptor tyrosine kinases and their ephrin ligands have intriguing expression patterns in cancer cells and tumour blood vessels, which suggest important roles for their bidirectional signals in many aspects of cancer development and progression. Eph gene mutations probably also contribute to cancer pathogenesis. Eph receptors and ephrins have been shown to affect the growth, migration and invasion of cancer cells in culture as well as tumour growth, invasiveness, angiogenesis and metastasis in vivo. However, Eph signalling activities in cancer seem to be complex, and are characterized by puzzling dichotomies. Nevertheless, the Eph receptors are promising new therapeutic targets in cancer.
Figures



References
-
- Pasquale EB. Eph-ephrin bidirectional signaling in physiology and disease. Cell. 2008;133:38–52. - PubMed
-
- Hirai H, Maru Y, Hagiwara K, Nishida J, Takaku F. A novel putative tyrosine kinase receptor encoded by the eph gene. Science. 1987;238:1717–1720. - PubMed
-
- Maru Y, Hirai H, Takaku F. Overexpression confers an oncogenic potential upon the eph gene. Oncogene. 1990;5:445–447. - PubMed
-
- Bartley TD, et al. B61 is a ligand for the ECK receptor protein-tyrosine kinase. Nature. 1994;368:558–560. - PubMed
-
- Pasquale EB. Eph receptor signalling casts a wide net on cell behaviour. Nat Rev Mol Cell Biol. 2005;6:462–475. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

